ISSCA to Conduct First Hands On Regenerative Medicine Training after Global Pandemic

The COVID19 pandemic has brought about a ‘new normal’ of social-distancing, closed businesses and self-isolation. After several months of inactivity brought about by this virus and the period of self-isolation, the International Society for Stem Cell Application is ready to begin the process of resuming our schedule of training courses.
Now, more than ever, interest in the immune system and its health has breached our collective consciousness, both phycicians and scientist are turning to regenerative medicine as a way to strengthen it. Recent breakthroughs in the field have led to a newly devised program– one with a proprietary protocol for strengthening the immune system, furthering a person’s ability to fight and prevent disease.
This inaugural training will take place in Washington over the course of two days, during which we will be training a group of five doctors in the latest regenerative medicine techniques. We will go over our traditional program providing physicians with all of the tools they will need to begin incorporating regenerative medicine into their own practice
The International Society for Stem Cell Application is an independent, non-profit organization whose objective is to become the gold standard of regenerative medicine. It accomplishes this through hosting conferences and specialized certification courses, publishing and sharing clinical research, and setting protocols in the field of regenerative medicine. Through this partnership, we are seeking to spread knowledge on the current applications of cellular therapies by building a platform for doctors to discuss and debate the newest advancements in the field. These conferences have been held in dozens of countries, and work to create a global network of regenerative medicine researchers and practitioners.
If you’d like to know more information about our program, or the courses that we offer around the world, please check out by visiting our website at: www.issca.us
About International Society for Stem Cells Applications
The International Society for Stem Cells Applications (ISSCA) is a multidisciplinary community of scientists and physicians, all of whom aspire to treat diseases and lessen human suffering through advances in science, technology, and the practice of regenerative medicine. Incorporated under the Republic of Korea as a non-profit entity, the ISSCA is focused on promoting excellence and standards in the field of regenerative medicine.
ISSCA bridges the gaps between scientists and practitioners in Regenerative Medicine. Their code of ethics emphasizes principles of morals and ethical conducts.
At ISSCA, their vision is to take a leadership position in promoting excellence and setting standards in the regenerative medicine fields of publication, research, education, training, and certification. ISSCA serves its members through advancements made to the specialty of regenerative medicine. They aim to encourage more physicians to practice regenerative
medicine and make it available to benefit patients both nationally and globally.
For more information, please visit https://www.issca.us/ or send an email to info@stemcellsgroup.com
- Published in Press Releases
GLOBAL STEM CELLS GROUP will host ISSCA Regenerative Medicine Training

After several months of silence due to the mandatory shutdown in place, the training division of Global Stem Cells Group –known as ISSCA Training– has officially announced the reopening of its regularly scheduled training dates Around the world
Miami, FL (PRUnderground) May 20th, 2020
“The future’s looking bright for our Regenerative MedicineTraining program, we’ve taken our course and updated it, reinforcing it with new information and quality protocols that’ll help doctors and their patients manage this ‘new normal’ as well as they can, and we’re very excited to be able to be bringing this new information to physicians around the world– but we’ve decided to start small for now, with the reinstatement of our headquarters’ operations,” — Benito Novas, CEO

Because of the growing interest in the industry , ISSCA Regenerative MedicineTraining has updated its program with new protocols focused on the optimization of the body at the cellular level. Notably, their upcoming June course will also include live practice patients in the treatments of autologous cell therapy, which includes PRP, adipose-derived stem cells, bone-marrow derived stem cells, exosomes, Umbilical Cord Blood Cells, and other novel techniques.
Stem Cell Training is a company that seeks to instruct medical professionals around the world on regenerative medicine techniques. They do this by sharing breakthrough scientific research on stem cell therapy and treatment, as well as developing and offering courses, seminars, and workshops based on this research.
About the Global Stem Cells Group:
Global Stem Cells Group is the parent company of six wholly owned operating companies dedicated entirely to stem cell research, training, products and solutions. Founded in 2010, the company combines dedicated researchers, physicians ,patient educators and solution providers with the shared goal of meeting the growing worldwide need for leading edge Regenerative Medicine treatments.
With a singular focus on this exciting new area of medical research, Global Stem Cells Group and its subsidiaries are uniquely positioned to become global leaders in cellular medicine.
Global Stem Cells Groups corporate mission is to make the promise of Regenerative medicine a reality for patients around the world. With each of GSCGs six operating companies focused on a separate research-based mission, the result is a global network of state-of-the-art regenerative technology.
- Published in Press Releases
Global Stem Cells Group Announces Launch of Vita Novas In-Home IV Infusion Service

Its no secret that a quality IV infusion can boost health and energy. Vita Novas makes this more simple than ever performing IV services than revitalize the body and help it regenerate, right at home.
Miami , Florida (PRUnderground) May 20th, 2020
The interest in boosting the immune system in a safe and effective way is a topic on more minds than ever. In exciting news, Global Stems Group has announced the launch of a new company, Vita Novas who make this both simple and dependable. Vita Novas offers an IV infusion packages are a mix of stem cells, essential fluids, electrolytes, vitamins and antioxidants as a preventive solution for having an optimal immune system. This IV infusion is delivered at home, in an office, or at a hotel, by certified healthcare professionals. With the global pandemic shutting doors of physicians for elective and health-enhancing non-vital procedures going to get IV treatments in most communities is difficult, if not impossible. Vita Novas is opening the doors for these life-enhancing treatments to be accesses in an easy and convenient way.
“The demand for preventive health services using IV infusion is greater than ever,” commented Benito Novas, CEO of the company. “What we are doing, by offering these at home and offices, is not just making things more convenient but also following social distancing requirements. Its truly making the right thing available at the right time and place.”
Vita Novas provides a teleconsultation with a Doctor to determine what meets a patient’s needs best. Three different vitamin and nutrient vitamin infusion packages are available.
The Lighting Package consists of two sessions of IV administration of vitamins (Vitamin C, B-Complex vitamins, and trace minerals), with a patient follow up recommending dietary changes and oral supplements to maintain good health.
The Shield Package is three sessions of IV administered (high dose Vitamin C, Methylcobalamin, Zinc sulphate, Glutathione and five billion flow exosomes). This has been shown have a powerful effect in regulating regenerative processes in the body and increase cell to cell communication. This has been used to treat COVID-19 patients with exciting results, something clearly at the front of people’s minds of all ages and lifestyles.
The Fighter Packages is the platinum offer from Vita Novas. This combines IV administration of high dose micronutrients (amino acids, glutathione), vitamins (B-Complex, methylcobalamin, Folic Acid, Vitamin C), and trace minerals (zinc, copper, magnesium) along with 30 million live stem cells in 5 sessions. This can modulate immunity and assist in the regeneration process of damaged or infected tissue in a very dramatic way.
Vita Novas is currently positioned to be able to launch the program in ten different countries with the number rising to over 25 within six months. The near and far future are bright for the company and its potentially life-changing services.
About the Global Stem Cells Group:
Global Stem Cells Group is the parent company of six wholly owned operating companies dedicated entirely to stem cell research, training, products and solutions. Founded in 2010, the company combines dedicated researchers, physicians ,patient educators and solution providers with the shared goal of meeting the growing worldwide need for leading edge Regenerative Medicine treatments.
With a singular focus on this exciting new area of medical research, Global Stem Cells Group and its subsidiaries are uniquely positioned to become global leaders in cellular medicine.
Global Stem Cells Groups corporate mission is to make the promise of Regenerative medicine a reality for patients around the world. With each of GSCGs six operating companies focused on a separate research-based mission, the result is a global network of state-of-the-art regenerative technology
- Published in Press Releases
Bogota Course Will Share the Latest in Stem Cell Treatment Protocols
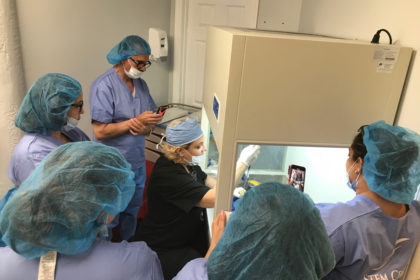
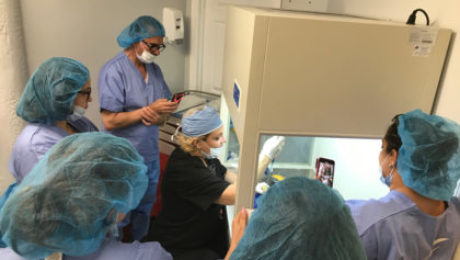
The two-day certification program is sponsored by ISSCA, will feature three topics new to the group’s Colombian training circuit
MIAMI LAKES, Florida— The International Society for Stem Cell Application (ISSCA), a global leader in stem cells research, applications, and education, has announced their latest certification program to be held in Bogota, Colombia on March 13-14, 2020.
The Stem Cells Certification course will feature a number of educational and hands-on sessions designed to give physicians the tools needed to integrate regenerative treatments into their practices. Registration for the two-day course is limited to eight participants to allow for intensive training and interaction.
During the two-day training, ISSCA faculty will share the latest stem cell protocols, including how to harvest and process stem cells derived from Stromal Vascular Faction (SVF) tissue and bone marrow.
ISSCA faculty will additionally introduce three new protocols never before covered in past courses held in Colombia. The new curriculum includes clinical applications of allogeneic stem cells; using GCell technology, a device that can process SVF without enzymatic compounds; and an introduction to NK cell protocols, the newest regenerative medicine treatment to fight cancer.
“Our two-day training in Bogota will provide physicians with an invaluable opportunity to receive intensive hands-on training and education in some of today’s most effective and cutting-edge stem cells protocols,” said Benito Novas, ISSCA VP of Public Relations. “Physicians will learn from global leaders in the stem cells industry and be able to take practical application strategies back with them to integrate into their own practices to help patients suffering from degenerative diseases.”To learn more about the ISSCA, visit www.issca.us. More information about the Bogota certification course can be found at http://cursocelulasmadre.com/entrenamiento-bogota-programa/
- Published in Press Releases
What is Exosome therapy?
Exosomes are extracellular vesicles (that’s particles that release naturally from a cell that cannot replicate) that are responsible for certain genetic information, otherwise known as Exosomes cell to cell communication. They transport molecules that are essential regulators of intracellular information between close and distant cells.
Exosomes play a vital role in the communication and rejuvenation of all the cells in our body. Despite not being a cell, Exosome has proven to be quite important in maintaining a healthy cellular terrain.
Exosomes are typically made by growing stem cells in culture. Taking the media where they grow and then getting rid of the stem cells. Due to the small size of Exosomes, the media is then ultra-centrifuged to concentrate the exosomes.
Exosomes contain different molecular constituents of their cell origin; these include proteins and RNA. Meanwhile, the Exosomal proteins composition varies with the cell and tissue of the origin; most Exsomes contains an evolutionarily conserved common set of protein molecules.
HOW TO ISOLATE EXOSOME FROM CULTURE STEM?
Exosomes can be isolated from culture stem cell using the method Ultracentrifugation. Often combined with sucrose density gradients, they float the relatively low-density Exosomes from other microvesicles. Cells and larger particles are removed by sequentially increasing the centrifugal forces. These procedures can take up to 30hours and requires an ultracentrifuge, extensive training and specialized equipment.
This method provides highly enriched exosomes but requires specialized training and equipment.
Now, this is how Exosomes works:
Exosomes carry genetic information. Protein and messenger RNA, they can relay this information, letting cells know when to and how to react. This Exosomes cell-to-cell communication is possible due to its unique shape and content.
Exosomes from young stem cells rejuvenate the older cells and assist in calming an overreactive immune system or modulating it to respond in a coordinated and more effective fashion.
WHAT IS EXOSOME THERAPY?
Exosome therapy is one of the hottest areas of regenerative medicine treatment. Researchers have given us valuable insight into the practical functionality of exosomes. While stem cells are usually responsible for the rejuvenation of older cells, they may not be able to supply all the information needed. So supporting Exosome function could have a greater positive effect, by providing a new piece of information to enhance the healing process.
Exosome therapy is a minimally invasive procedure commonly used in orthopedic injuries, for anti-aging and some other degenerative diseases. Exosomes treatment contains growth factors, messenger RNA, micro RNA, cytokines and other biologically active molecules in conjunction with stem cell therapy to speed up healing benefit.
Exosome therapy can be injected into the affected area like in orthopedic injuries or given intravenously for anti-aging.
This Exosome injection is administered directly into the affected area, and the dosing for every patient varies, and there is no set protocol.
Exosomes treatment is gaining popularity recently, due to its ability to transfer molecules from one cell to another via membrane vesicle, therefore influencing the immune system, such as dendrite cells and B cells.
What is exosome therapy used for?
HAIR LOSS: Exosomes injection can be used to regrow hair loss due to its growth factor content. When injected, Exosome will trigger healing, cell stimulation and natural regeneration of these hair follicles. Exosome hair loss therapy is cutting-edge in hair restoration. According to research, there is a positive relationship between Exosomes and Hair loss. Exosomes are ideal for people with thinning hair, excessive shedding or hair loss proper. This is because Exosomes are regenerative cells that can heal, repair, stimulate and restore cells and tissues.
LYME DISEASE: Exosomes may help to regulate processes in the body, and may also be beneficial to patients with Lyme disease. Lyme disease is a very complex disease, which compromises the immune system. Many Lyme patients suffer from dysfunction of the mast cells, increasing their inflammatory response and metabolic function. Incorporating Exosomes treatment in their treatment regimen may help break the inflammatory response and provide the body with necessary cellular information to facilitate healing.
ARTHRITIS: Arthritis as we all know, is the joint inflammation resulting from an autoimmune disease. Although various types of treatment are available to alleviate symptoms, no known therapy has been confirmed effective in reversing the disease progression.
In the field of regenerative medicine today, with the discovery of extracellular microvesicles, especially exosomes, many researchers have been able to offer a more exciting alternative on this subject matter. Exosome arthritis therapy is believed to play a more substantial role in bone and cartilage remodeling.
In a nutshell, Exosomes provides enormous potential in the field of regenerative medicine. You can advance your understanding of Exosomes and its relationship with stem cell in this course. This is an online-course https://www.cellulartherapycourse.com/ where physicians and medical personnel can get insight and deep understanding of this novel science (Exosome).
- Published in Corporate News / Blog
How to boost your immune system to fight cancer?
The human immune system helps to protect the body against illness and infection caused by bacteria, viruses, fungi, and parasites. This process is achieved by a collection of reactions and responses made by the body to damage these infected cells. It is also called immune responses.
This immune system is very crucial in cancer patients to help fight cancer, it dictates this cancer as abnormal and destroys it, but this is not enough to rid of cancer altogether. In most cases, cancer will weaken this immune system because it will spread into the bone marrow that produces blood cells that fights against infections and cause the bone marrow to decrease in its blood cell production.
When this immune system is unable to recognize these cells as abnormal, cellular growth will occur, causing uncontrolled growth and division of cells, that will, in turn, lead to cancer.
Cancer has been a serious and sensitive talk for both patients and doctors. Being diagnosed with cancer is frightening and tormenting, but due to the latest treatment method, recovering from this disease is increasingly possible. Living with cancer often brings an initial psychological crisis, but surprisingly it does not only affect you but your family and others close to you.
Cancers, especially genetic cancer can’t be prevented, so it is essential to be proactive about your health. If you’re diagnosed with cancer, some individual lifestyles that will help improve your cancer care, which includes managing stress. Reducing your stress level can help you to maintain physical and mental stress; you can use relaxation techniques such as meditation and yoga. Getting enough sleep is also crucial for patients living with cancer; this will help improve their health, mood, and coping ability. Also, it is best the limit their exposure to toxins that can increase one’s risk of being exposed to other deadly diseases.
A healthy feeding habit is critically important and can help manage cancer side effects, improve health, and quick recovery. Some tips to help you develop a good and healthy feeding diet include:
- Consuming assorted types of vegetables in all your meals. Vegetables are essential and should not just be a side dish but the centerpiece of your meal.
- Consuming foods rich in fiber like grains, beans, peas, nuts, and seeds.
- Consuming less red meat, like pork, lamb, goat, and bison.
- The presence of probiotic and prebiotic foods helps to support a healthy gut. Probiotic foods include yogurt or other fermented vegetables, and prebiotic foods include raw or cooked onions, dandelion greens, and legumes.
- Avoid processed and canned foods
- Avoid foods high in calories and low in nutrients like fruit-flavored drinks, sodas, candies, and sweets.
- Lastly, cut down on the excessive intake of alcohol.
Some other routines can help improve cancer care like regular exercise. Regular exercise helps to control fatigue, muscle tension, and anxiety in those with cancer, and it is essential, especially during and after cancer treatment, so keep fit and avoid adding unnecessary weight.
CANCER TREATMENT
There are many cancer treatment options for different types of cancers. Cancer treatments depend on the type and how to advance it is. Some patients with cancer will only have one treatment option, while others will end up with a combination of more that one treatments technique depending on the type and stage of cancer.
Curing cancer is one of the significant challenges of the 21st century. However, few advances to tame the immune system are getting closer to a future where cancer can be a curable disease.
Although cancer treatment is dependent on the stage and type, we need a wide range of therapies that work on cellular level like NK cell and stem cell therapies, wide enough to cover the whole spectrum of cancer. Cancer therapies involving the immune system is believed to be a milestone for cancer treatment advances, and the development of multiple immune cells as a therapeutic tool.
Our knowledge of cancer has dramatically improved in recent years. And it seems increasingly evident that there has been a surge of new technologies, and these technologies could make a great impact on the way we treat cancer, taking us closer to finally curing this deadly disease.
One of these promising cancer treatment advances is the natural killer (NK) cell-based immunotherapy.
NK CELLS
Natural killer cells have emerged as the newest promising therapeutic approach to cancer. Our understanding of the biological processes that take place in cancer is increasing rapidly, leading to this new type of targeted treatment and therapeutic approach. It is hard to overstate the possible importance of NK cells in the future of cancer. Modern researches have shown that NK cells are a type of treatment that stimulates a persons’ immune system to fight cancer, and this may give room for a greater number of chances to beat cancer for good. NK cells are also known for their ability to target cancer stem cells making these cancer stem cells visible to the immune system.
NK cells was first identified in the 1970s as a unique lymphocyte subset that can recognize and kill abnormal cells without prior sensitization of specific tumor antigen, thus preventing the growth of many cancers. During the late 1980s, something was observed, and this was that NK cells could destroy a lymphoma cell line that had lost MHC class I surface molecules while the original MHC class I+ cells were resistant to lysis, which led to the formation of the “missing self-hypothesis” which states that NK cells can sense the absence of “self” MHC class-I molecule on target cells. In recent years, the hypothesis was confirmed by the inhibitory and activating NK cell receptors.
This discovery indicates that autologous cells are not killed by NK cells, thanks to an appropriate expression of all self-HLA alleles, do not kill an autologous cell. In contrast, a wide spec of cancer types can be killed due to the loss of HLA molecules and to the expression/overexpression of ligands for NK cell activating receptors.
Now, this is how NK therapy works
NK cells firsts recognize the tumor cells via stress or danger signals from their sensors.
Then, the Activated NK cells directly decimate the target tumor cells through at least four mechanisms. These mechanisms are cytoplasmic granule release, death receptor-induced apoptosis, effector molecule production, or ADCC.
Furthermore, NK cells act as regulatory cells when reciprocally interact with DCs to improve their antigen uptake and presentation. This action helps in facilitating the generation of antigen-specific CTL responses that is vital in dictating foreign invasion and tumor cells.
Also, by producing cytokines such as IFN-γ, activated NK cells induce CD8+ T cells to become CTLs. Activated NK cells can also help in the differentiation of CD4+ T cells toward a Th1 response and promote CTL differentiation. Cytokines produced by NK cells might even have the unique ability to regulate antitumor cells, making them more proactive.
Accordingly, The cytokine gene transfer approach induces NK cell proliferation, and this increases survival capacity. Further enhancing their activation and also making them more potent in malignant cell multiplication dictation.
The NK cell scientist has assured that by using NK cell lines, modifying genes such as IL-2, IL-12, IL-15, and stem cell factor (SCF). They have been demonstrated the ability of the NK to restore their cytotoxic capacity as well as increase their proliferative rate, survival ability, and in vivo antitumor activity. However, the specificity of NK cells is still limited, and studies are even passing through phases. The approach focuses on retargeting NK cells to tumor cells by gene transfer of chimeric tumor-antigen specific receptors.
NK therapy is promising research; it raises the prospect of “one-end-solution” to many different types of cancers across the Globe. If the studies and experiments work out pretty well, the NK cells will be able to increase the activity of CD4+, CD19, and other important disease-fighting cells of the body.
There is no doubt that this an unimaginable feat, both in advancing our basic knowledge about regenerative medicine and for the possibility of future cancer treatment advances.
- Published in Corporate News / Blog
Can Stem Cells treatment Regrow the knee cartilage?
Some people believe that stem cell does all of this but studies have shown that It’s unlikely only in a few unique cases. You can observe minimal growth a year after the patient took treatment, but this doesn’t mean replacement of the cartilage.
The cartilage has a reduced regenerative capacity, and current and present pharmacological medications only offer symptomatic pain relief. Osteoarthritis patients that respond poorly to conventional therapies are ultimately treated with surgical procedures to promote cartilage repair by implantation of artificial joint structures (arthroplasty) or total joint replacement (TJR). Surgery has been the last resort for serious cartilage problems.
In the last two decades, stem cells derived from various tissues with varying differentiation and tissue regeneration potential have been used for the treatment of osteoarthritis, damage to bones and others either alone or in combination with natural or synthetic scaffolds. The stem cells derived from these tissues primarily aid cartilage repair. Although stem cells can be differentiated into chondrocytes in vitro or aid cartilage regeneration in vivo, their potential for Osteoarthritis management remains limited as cartilage regenerated by stem cells fails to fully recapitulate the structural and biomechanical properties of the native tissue. It isn’t easy for the cartilage to regrow and assume its original biomechanical and structure form.
Apparently, Due to the limited intrinsic capacity of resident chondrocytes to regrow the lost cartilage post-injury, stem cell-based therapies have been proposed as a novel therapeutic approach for cartilage repair.
Also, stem cell-based therapies using mesenchyme stem cells (MSCs) or induced pluripotent stem cells (iPSCs) have been used successfully in clinical and preclinical situations.
Part of the issues associated with Mesenchyme stem cells can be averted by using iPSCs. iPSCs are an ideal patient-specific unlimited cell source for autologous tissue regeneration. With the Promising in vitro; studies have shown that vitro results have already been demonstrated in the cartilage engineering field for iPSCs. These were generated from various cell types.
What Is Cartilage and How Does It Get Damaged?
Cartilage is a connective tissue in the human body and body of other animals. In our joints, we have a few kinds of cartilage, but most often people refer to the smooth lining of a joint called articular or hyaline cartilage. This kind of cartilage gives rise to a soft layer of cushion on the end of a bone at the joint. The cushion is essential for balance, mechanical functions and athletics. This tissue of the cartilage is very strong, yet it can compress, readjust and absorb varying degrees of energy. It is also very slippery, smooth and flexible and these features allow the joint to glide effortlessly through a broad range of physical motions of any kind.
When joint cartilage is not working correctly or damaged, this smooth-cushioning-layer can be worn away, and this becomes a problem. In the case of traumatic injuries, sometimes a sudden force causes the cartilage to break off or poorly become damaged, exposing the underlying bone of the body. In the case of osteoarthritis (also called degenerative/wear-and-tear arthritis), over time that smooth layer can wear thin and uneven. Aging can also cause the cartilage to break off and certain life factors and diseases too, e.g. autoimmune diseases.
Eventually, as that cushion of the bones wears away, joint movements can become inflexible, stiff and painful on one or both legs (bones). Joints can even become inflamed and swollen. And as all these conditions, typically causes pain and limitations in activity become problematic. The action or activities that involve these bones leads to crushing pain and discomfort, depending on the severity of the case. Almost all activities involve the movement of bones; hence this condition is not an easy one.
There are some treatments for cartilage damage and arthritis. Although there some medicines, most of these treatments are focused either on relieving symptoms by smoothing down the damaged cartilage or concentrate on replacing the joint surface with an artificial implant. The later is for end-stage conditions, and the artificial plane is procedures such as knee replacement or hip replacement surgery.
How Can Stem Cells Help?
Stem cells are specialized cells that can multiply reform and develop into different types of tissue. In the developmental stages of a fetus, stem cells are plentiful and surplus. However, in adulthood, stem cells are restricted to specific tasks of regenerating a few types of cells, such as blood cells and liver cells in some cases of damage. There are almost no stem cells found in cartilage tissue, and therefore there is little to no capacity to heal or regrow new cartilage. For adults, the ability to regrow new cartilage is even more difficult due to age and lack of stem cells in the cartilages.
Most often, in the setting of orthopedic surgery and joint problems, stem cells are obtained from adult stem cell sources. The primary sources are bone marrow and fatty tissue. These stem cells can develop into cartilage cells, called chondrocytes.
They also exhibit some other helpful qualities by stimulating the body to reduce inflammation, stimulate cell repair, and improve blood flow. This process is caused by the secretion of cellular signals and growth factors to stimulate the body to initiate healing processes.
Once stem cells have been obtained, they need to be delivered to the area of the cartilage that damaged. One option is to inject the stem cells into the joint. There have been many studies investigating just this, and some data shows improvement in symptoms. How much of this improvement is the result of new cartilage growth versus other effects of stem cells (the healing properties listed above, including the anti-inflammatory effects) is unknown.
There is a challenge with giving stem cell injection. The problem with just injecting stem cells is that cartilage is a complex tissue that is comprised of more than only cells hence this can pose a challenge because the stem can’t regenerate all the things in the cartilage.
To regrow the cartilage, the complex tissue structure and biomechanics of cartilage must also be reconstructed to its former status. Cartilage can often /described as having a scaffold-like structure that is composed of water, cells, collagen, and proteoglycans, and infection-fighting antibodies. Injecting just the stem cells is thought to be less effective in stimulating the formation of the entire cartilage structure hence the challenge.
Some studies are investigating the types of 3-dimensional tissue scaffolds engineered to have a cartilage-like structure. The stem can then be injected into the scaffold, in hopes of better restoring a healthy type of cartilage. Three-dimensional printing is becoming an exciting part of this type of research. If everything works out as expected, the cartilage reconstruction could be achieved to a very high percentage.
How do stem cells work?
Necessarily, stem cells are progenitor cells which are capable of regeneration and differentiation into a wide range of specialized cell types. Once injected, stem cells follow inflammatory signals from damaged tissues and have multiple ways of repairing these damaged areas. It works as though the part is developing new; like what is seen during a child’s development.
The mesenchyme stem cells (MSCs) we are using are considered to be multipotent (they can transform into different cell types but cannot form an organ) but not pluripotent. In the body, these cells Do NOT function by transforming into different cell types or tissues.
They act via anti-inflammatory activity, immune modulating capacity, and the ability to stimulate regeneration. We go through a very high thorough screening process to find cells that we know have the best anti-inflammatory activity, the best immune modulating capacity, and the best ability to stimulate regeneration process on the tissue with damage.
ISSCA (International Society for Stem Cells Applications) www.issca.us
This is a business located in Miami, FL, where people around the world come to take a certification in the newest Stem Cells Protocols.
Some organizations have put in efforts to help discover some solutions in stem medicine. International Society for Stem Cell Application (ISSCA ) is one of the leading associations in setting standards and promoting excellence in the field of Regenerative Medicine, researches, publications related education, certification, research and publications.
The ISSCA is a unique-multidisciplinary community of physicians, stem specialist and scientists with a mission to advance the science, technology and practice of Regenerative Medicine. Their aim is to treat disease and lessen human suffering. ISSCA generally advances the specialty of Regenerative Medicine and serves its members.
The ISSCA provides certifications and standards in the practice of Regenerative Medicine as a medical specialty.
Although the expectation on this stem cell course is yet to be achieved; however, this is a part of medicine that can offer one-end-solution to various bone and body problems.
With the recent high-tech studies, efforts and dynamics, stem cell treatment can be a breakthrough in the future as its perspectives are very promising and unique. It is also not dangerous on the long-run.
- Published in Corporate News / Blog
Clinical trials on NK cells give hope for many people Who are suffering from cancer.
Induced pluripotent stem cells (also known as iPS cells or iPSCs) are a type of pluripotent stem cell that can be generated directly from a somatic cell. Pluripotent stem cells hold promise in the field of regenerative medicine. Because they can propagate indefinitely, as well as give rise to every other cell type in the body (such as neurons, heart, pancreatic, and liver cells), they represent a single source of cells that could be used to replace those lost to damage or disease.
Natural killer cells are the type of cytotoxic lymphocyte critical to the innate immune system. The role NK cells play is analogous to that of cytotoxic T cells in the vertebrate adaptive immune response. NK cells provide rapid responses to virus-infected cells, acting at around three days after infection, and respond to tumor formation.
Typically, immune cells detect the major histocompatibility complex (MHC) presented on infected cell surfaces, triggering cytokine release, causing apoptosis. NK cells are unique, however, as they can recognize stressed cells in the absence of antibodies and MHC, allowing for a much faster immune reaction.
Clinical Trial on NK cells
In a first clinical trial, a natural killer cell immunotherapy derived from induced pluripotent stem cells is being tested for safety in 64 patients with a variety of solid tumors. The first subjects used for the study received the cells in February at the University of California, San Diego (UCSD) Moores Cancer Center and MD Anderson Cancer Center.
This study is targeting late-stage cancer patients with solid tumors, including lymphoma, colorectal cancer, and breast cancer. The FT500 NK cells do not undergo any further alterations and after their derivation from the induced pluripotent stem cells (iPSCs), offering the possibility of a quicker, ready-made treatment.
Human embryonic stem cells induced iPSCs
Human embryonic stem cells (hESCs) and induced pluripotent stem cells (iPSCs) provide an accessible, genetically tractable, and homogenous starting cell population to efficiently study human blood cell development. These cell populations provide platforms to develop new cell-based therapies to treat both malignant and nonmalignant hematological diseases.
The NK cells are immune cells in the same family as T and B cells and are very good at targeting cancer cells for destruction. Some Laboratory experiments have shown they do so by attacking cells that have lost their significant self-recognition signals that tell the immune system not to attack. This is the phenomenon that can happen among cancer cells but not to healthy cells. Experts are not sure how many cancer cells lose that signal. Researchers are hopeful that the clinical trial can help determine which cancer patients could benefit the most from NK cell treatment.
iPS Clone
The ability to induce pluripotent stem cells from committed, human somatic cells provides tremendous potential for regenerative medicine. However, there is a defined neoplastic potential inherent to such reprogramming that must be understood and may offer a model for critical understanding events in the formation of the tumor. Using genome-wide assays, we identify cancer-related epigenetic abnormalities that arise early during reprogramming and persist in induced pluripotent stem cell (iPS) clones. These include hundreds of abnormal gene silencing events, patterns of aberrant responses to epigenetic-modifying drugs resembling those for cancer cells, and presence in iPS and partially reprogrammed cells of cancer-specific gene promoter DNA methylation alterations.
Progress in adoptive T-cell therapy for cancer and infectious diseases is hampered by the lack of readily available antigen-specific, human T lymphocytes. Pluripotent stem cells could provide an estimable source of T lymphocytes, but the therapeutic potential of human pluripotent stem cell-derived lymphoid cells generated to date remains uncertain.
Modification of T cells
Recently, some Approved cell therapies for Cancer also rely on modifying T cells, in those cases to produce cancer cell–binding chimeric antigen receptors (CARs), and have been effective in treating certain cancers such as leukemia.
Application of CAR T-Cell Therapy in Solid tumours
The Car T technology has wowed the field by all but obliterating some patients’ blood cancers, but solid malignancies present new challenges.
Therapies that contains such chimeric antigen receptor (CAR) T cells have been approved for some types of so-called liquid cancers of the blood and bone marrow, large B-cell lymphoma and B-cell acute lymphoblastic leukemia. But the approach has not had as much success for solid tumors.
Serious research into the therapy for brain cancer started almost 20 years ago after cancer biologist WaldemarDebinski, then at Penn State, discovered that the receptor for the immune signaling molecule interleukin 13 (IL-13) was present on glioblastomas, but not on healthy brain tissue. The receptor thus seemed like an excellent target to home in on cancer cells while sparing healthy ones. The CAR spacer domain that spans the immune cells’ membranes and its intracellular co-stimulatory areas, as well as the process used to expand cells outside the body, to boost the T cells’ activity.
CAR T- A Safer Cell Therapy
While managing CAR T-cell therapy toxicity could help keep already-designed treatments on their march to the clinic, many immunotherapy companies are also working to develop a new generation of inherently safer therapies, yet just as efficient. A crucial part of achieving this goal will be improving CAR specificity for target cells. With current treatments, the destruction of normal cells is often an unavoidable side effect when healthy tissue carries the same antigens as tumors; noncancerous B cells, for example, are usually casualties in CD19-targeted therapies.
CAR T delivery is a non-easy factor in the treatment of solid tumors and other unknown forms of tumors. With the non-solid cancers, cells are administered by a blood infusion, and once in circulation, the CAR T can seek out and destroy the rogue cells. For solid tumors, it’s not so simple.
The main drawback of taking cells from a patient and developing them into a cellular immunotherapy product is that the process can take weeks.
Patel tells The Scientist “But for the majority of patients who may not be a candidate or may not have time to wait for such an approach, the idea that there’s off-the-shelf immunotherapy that could potentially as a living drug act against their cancer, I think is a fascinating concept,”
https://www.issca.us/#upcomingevents
- Published in Corporate News / Blog
Stem cell treatment could offer one-end-solution to Diabetes
Insulin-producing cells grown in the lab could provide a possible cure for the age-long disease (diabetes).
Type 1 diabetes is an auto¬immune disease that wipes out insulin-producing pancreatic beta cells from the body and raises blood glucose to dangerously high levels. These high levels of Blood sugar level can be even fatal. Patients are being administered insulin and given other medications to maintain blood sugar level. To those who cannot maintain their blood sugar level, they are given beta-cell transplants but to tolerate beta cell transplants; patients have to take immunosuppressive drugs as well.
A report by a research group at Harvard University tells us that they used insulin-producing cells derived from human embryonic stem cells (ESCs) and induced pluripotent stem cells to lower blood glucose levels in mice. Nowadays, many laboratories are getting rapid progress in human stem cell technology to develop those cells that are functionally equivalent to beta-cells and the other pancreatic cell types. Other groups are developing novel biomaterials to encapsulate such cells and protect them against the immune system without the need for immunosuppressant.
Major pharmaceutical companies and life sciences venture capital firms have invested more than $100 million in each of the three most prominent biotechnological industries to bring such treatments into clinical use:
- Cambridge
- Massachusetts–based companies Semma Therapeutics
- Sigilon Therapeutics, and ViaCyte of San Diego
Researchers of UC San Francisco have transformed human stem cells into mature insulin-producing cells for the first time, a breakthrough in the effort to develop a cure for type-1 (T1) Diabetes. Replacing these cells, which are lost in patients with T1 diabetes, has long been a dream of regenerative medicine, but until now scientists had not been able to find out how to produce cells in a lab dish that work as they do in healthy adults.
What is T1 diabetes?
T1 diabetes is an autoimmune disorder that destroys the insulin-producing beta cells of the pancreas, typically in childhood. Without insulin’s ability to regulate glucose levels in the blood, spikes in blood sugar can cause severe organ damage and eventually death. The condition can be managed by taking regular shots of insulin with meals. However, people with type 1 diabetes still often experience serious health consequences like kidney failure, heart disease and stroke. Patients facing life-threatening complications of their condition may be eligible for a pancreas transplant from a deceased donor, but these are rare, and they are supposed to wait a long time.
Researchers have just made a breakthrough that might one day make these technologies obsolete, by transforming human stem cells into functional insulin-producing cells (also known as beta cells) – at least in mice.
“We can now generate insulin-producing cells that look and act a lot like the pancreatic beta cells you and I have in our bodies,” explains one of the team, Matthias Hebrok from the University of California San Francisco (UCSF).
“This is a critical step towards our goal of creating cells that could be transplanted into patients with diabetes.”
Type-1 diabetes is characterized by a loss of insulin due to the immune system destroying cells in the pancreas – hence, type 1 diabetics need to introduce their insulin manually. Although this is a pretty good system, it’s not perfect.
Making insulin-producing cells from stem cells
Diabetes can be cured through an entire pancreas transplant or the transplantation of donor cells that produce insulin, but both of these options are limited because they rely on deceased donors. Scientists had already succeeded in turning stem cells into beta cells, but those cells remained stuck at an early stage in their maturity. That meant they weren’t responsive to blood glucose and weren’t able to secrete insulin in the right way.
Scientists at the University of California San Francisco made a breakthrough in the effort to cure diabetes mellitus type 1.
For the first time, researchers transformed human stem cells into mature insulin-producing cells, which could replace those lost in patients with the autoimmune. There is currently no known way to prevent type-1 (T1) diabetes, which destroys insulin production in the pancreas, limits glucose regulation, and results in high blood sugar levels. The condition can be managed with regular shots of insulin, but people with the disease often experience serious health complications like kidney failure, heart disease, and stroke.
“We can now generate insulin-producing cells that look and act a lot like the pancreatic beta cells you and I have in our bodies,” according to Matthias Hebrok, senior author of a study published last week in the journal Nature Cell Biology.
“This is a critical step toward our goal of creating cells that could be transplanted into patients with diabetes,” Hebrok, director of the UCSF Diabetes Center, said in a statement.
Islets of Langerhans are groupings of cells that contain healthy beta cells, among others. As beta cells develop, they have to separate physically from the pancreas to form these islets.
The team artificially separated the pancreatic stem cells and regrouped them into these islet clusters. When they did this, the cells matured rapidly and become responsive to blood sugar. In fact, the islet clusters developed in ways “never before seen” in a lab. After producing these mature cells, the team transplanted them into mice. Within days, the cells were producing insulin similar to the islets in the mice. While the study has been successful in mice, it still needs to go through more rigorous testing to see if it would work for humans as well. But the research is up-and-coming. “We can now generate insulin-producing cells that look and act a lot like the pancreatic beta cells you and I have in our bodies. This is a critical step towards our goal of creating cells that could be transplanted into patients with diabetes,” He said.
“We’re finally able to move forward on several different fronts that were previously closed to us,” he added. “The possibilities seem endless.”
Basic research keeps elucidating new aspects of beta cells; there seem to be several subtypes, so the gold standard for duplicating the cells is not entirely clear. Today, however, there is “a handful of groups in the world that can generate a cell that looks like a beta cell,” says Hebrok, who currently acts as scientific advisor to Semma and Sigilon, and has previously advised ViaCyte. “Certainly, companies have convinced themselves that what they have achieved is good enough to go into patients.”
The stem cell reprogramming methods that the three companies use to prompt cell differentiation create a mixture of islet cells. Beta cells sit in pancreatic islets of Langerhans alongside other types of endocrine cells. Alpha cells, for example, churn out glucagon, a hormone that stimulates the conversion of glycogen into glucose in the liver and raises blood sugar. Although the companies agree on the positive potential of islet cell mixtures, they take different approaches to developing and differentiating their cells. Semma, which was launched in 2014 to commercialize the Harvard group’s work and counts Novartis among its backers, describes its cells as fully mature, meaning that they are wholly differentiated into beta or other cells before transplantation. “Our cells are virtually indistinguishable from the ones you would isolate from donors,” says Semma chief executive officer BastianoSanna
To get around the donor problem, researchers, including the team at UCSF has been working on nudging stem cells into becoming fully-functional pancreatic beta cells for the last few years. Still, there have been some issues in getting them all the way there.
“The cells we and others were producing were getting stuck at an immature stage where they weren’t able to respond adequately to blood glucose and secrete insulin properly,” Hebrok said.
“It has been a major bottleneck for the field.”
“We’re finally able to move forward on a number of different fronts that were previously closed to us,” Hebrok added. “The possibilities seem endless.”
Regardless of starting cell type, the companies say they are ready to churn out their cells in large numbers. Semma, for example, can make more islet cells in a month than can be isolated from donors in a year in the United States, Sanna says, and the company’s “pristine” cells should perform better than donor islets, which are battered by the aggressive techniques required for their isolation.
As these products, some of which have already entered clinical trials, move toward commercialization, regulatory agencies such as the US Food and Drug Administration (FDA) and the European Medicines Agency have expressed concern about the plasticity of the reprogrammed cells. All three firms subject their cells to rigorous safety testing to ensure that they don’t turn tumorigenic. Before successful trials, companies won’t know the dose of beta cells required for a functional cure, or how long such “cures” will last before needing to be boosted. There’ll be commercial challenges, too: while the companies are investing heavily to develop suitable industrial processes, all acknowledge that no organization has yet manufactured cell therapies in commercial volumes.
Nevertheless, there’s growing confidence throughout the field that these problems will be solved, and soon. “We have the islet cells now,” says Alice Tomei, a biomedical engineer at the University of Miami who directs DRI’s Islet Immuno-engineering Laboratory.
“These stem cell companies are working hard to try to get FDA clearance on the cells.”
Protecting stem cell therapies from the immune system
Whatever the type of cell being used, another major challenge is delivering cells to the patient in a package that guards against immune attack while keeping cells fully functional. Companies are pursuing two main strategies:
- Microencapsulation, where cells are immobilized individually or as small clusters, in tiny blobs of a biocompatible gel.
- Macroencapsulation, in which greater numbers of cells are put into a much larger, implantable device.
ViaCyte, which recently partnered with Johnson & Johnson, launched its first clinical trial in 2014. The trial involved a micro-encapsulation approach that packaged up the company’s partially differentiated, ESC-derived cells into a flat device called the PEC-Encapsulation. About the size of a Band-Aid, the device is implanted under the skin, where the body forms blood vessels around it. “It has a semipermeable membrane that allows the free flow of oxygen, nutrients, and glucose,” says ViaCyte’s chief executive officer, Paul Laikind. “And even proteins like insulin and glucagon can move back and forth across that membrane, but cells cannot.”
The trial showed that the device was safe, well-tolerated, and protected from the adaptive immune system—and that some cells differentiated into working islet cells. But most cells didn’t engraft effectively because a “foreign body response,” a variant of wound healing, clogged the PEC-Encap’s membrane and prevented vascularization. ViaCyte stopped the trial and partnered with W. L. Gore & Associates, the maker of Gore-Tex, to engineer a new membrane. “With this new membrane,” says Laikind, “we’re not eliminating that foreign body response, but we’re overcoming it in such a way that allows vascularization to take place.” The company expects to resume the trial in the second half of this year, provided it receives the green light from the FDA.
Semma is also developing macro¬-encapsulation methods, including a very thin device that in prototype form is about the size of a silver dollar coin. The device is “deceptively simple, but it allows us to put [in] a fully curative dose of islets,” Sanna says.
Semma is also investigating microencapsulation alternatives. At the same time, the company is advancing toward clinical trials using established transplantation techniques to administer donated cadaver cells to high-risk patients who find it particularly difficult to control their blood glucose levels. These cells are infused via the portal vein into the liver, and patients take immunosuppressive drugs to prevent rejection.
Sigilon is working on its microencapsulation technology. Launched in 2016 on the back of work by the labs of Robert Langer and Daniel Anderson at MIT, the company has created 1.5-millimeter gel-based spheres that can hold between 5,000 and 30,000 cells (Nat Med, 22:306–11, 2016). Each sphere is like a balloon, with the outside chemically modified to provide immune-protection, says Sigilon chief executive officer Rogerio Vivaldi. “The inside of the balloon is full of a gel that creates almost a kind of a matrix net where the cells reside.”
In 2018, shortly after partnering with Eli Lilly, Sigilon and collaborators published research showing that islet cells that were encapsulated in gel spheres and transplanted into macaques remained functional for four months. The company has not disclosed a time frame for a type 1 diabetes trial “but we’re moving pretty quickly,” says chief scientific officer David Moller.
Conclusion
To conclude, all three firms hope to extend their work to treat some of the 400 million people worldwide with type 2 diabetes, many of them eventually benefit from insulin injections. The recent endorsements from big Pharmaceutical underline the real progress in beta-cell transplants, says Aaron Kowalski, a molecular geneticist and chief executive officer at JDRF, a foundation based in New York that has funded research at ViaCyte and academic labs whose work has been tapped by Semma and Sigilon. “These companies all realize that if they don’t do it, somebody else will. It’s hard to predict exactly when, but somebody is going to make this work.”
- Published in Corporate News / Blog






![Secondary-Diabetes[1]](https://stemcellcenter.net/wp-content/uploads/2020/02/Secondary-Diabetes1-460x260_c.png)