Stem cell treatment could offer one-end-solution to Diabetes
Insulin-producing cells grown in the lab could provide a possible cure for the age-long disease (diabetes).
Type 1 diabetes is an auto¬immune disease that wipes out insulin-producing pancreatic beta cells from the body and raises blood glucose to dangerously high levels. These high levels of Blood sugar level can be even fatal. Patients are being administered insulin and given other medications to maintain blood sugar level. To those who cannot maintain their blood sugar level, they are given beta-cell transplants but to tolerate beta cell transplants; patients have to take immunosuppressive drugs as well.
A report by a research group at Harvard University tells us that they used insulin-producing cells derived from human embryonic stem cells (ESCs) and induced pluripotent stem cells to lower blood glucose levels in mice. Nowadays, many laboratories are getting rapid progress in human stem cell technology to develop those cells that are functionally equivalent to beta-cells and the other pancreatic cell types. Other groups are developing novel biomaterials to encapsulate such cells and protect them against the immune system without the need for immunosuppressant.
Major pharmaceutical companies and life sciences venture capital firms have invested more than $100 million in each of the three most prominent biotechnological industries to bring such treatments into clinical use:
- Cambridge
- Massachusetts–based companies Semma Therapeutics
- Sigilon Therapeutics, and ViaCyte of San Diego
Researchers of UC San Francisco have transformed human stem cells into mature insulin-producing cells for the first time, a breakthrough in the effort to develop a cure for type-1 (T1) Diabetes. Replacing these cells, which are lost in patients with T1 diabetes, has long been a dream of regenerative medicine, but until now scientists had not been able to find out how to produce cells in a lab dish that work as they do in healthy adults.
What is T1 diabetes?
T1 diabetes is an autoimmune disorder that destroys the insulin-producing beta cells of the pancreas, typically in childhood. Without insulin’s ability to regulate glucose levels in the blood, spikes in blood sugar can cause severe organ damage and eventually death. The condition can be managed by taking regular shots of insulin with meals. However, people with type 1 diabetes still often experience serious health consequences like kidney failure, heart disease and stroke. Patients facing life-threatening complications of their condition may be eligible for a pancreas transplant from a deceased donor, but these are rare, and they are supposed to wait a long time.
Researchers have just made a breakthrough that might one day make these technologies obsolete, by transforming human stem cells into functional insulin-producing cells (also known as beta cells) – at least in mice.
“We can now generate insulin-producing cells that look and act a lot like the pancreatic beta cells you and I have in our bodies,” explains one of the team, Matthias Hebrok from the University of California San Francisco (UCSF).
“This is a critical step towards our goal of creating cells that could be transplanted into patients with diabetes.”
Type-1 diabetes is characterized by a loss of insulin due to the immune system destroying cells in the pancreas – hence, type 1 diabetics need to introduce their insulin manually. Although this is a pretty good system, it’s not perfect.
Making insulin-producing cells from stem cells
Diabetes can be cured through an entire pancreas transplant or the transplantation of donor cells that produce insulin, but both of these options are limited because they rely on deceased donors. Scientists had already succeeded in turning stem cells into beta cells, but those cells remained stuck at an early stage in their maturity. That meant they weren’t responsive to blood glucose and weren’t able to secrete insulin in the right way.
Scientists at the University of California San Francisco made a breakthrough in the effort to cure diabetes mellitus type 1.
For the first time, researchers transformed human stem cells into mature insulin-producing cells, which could replace those lost in patients with the autoimmune. There is currently no known way to prevent type-1 (T1) diabetes, which destroys insulin production in the pancreas, limits glucose regulation, and results in high blood sugar levels. The condition can be managed with regular shots of insulin, but people with the disease often experience serious health complications like kidney failure, heart disease, and stroke.
“We can now generate insulin-producing cells that look and act a lot like the pancreatic beta cells you and I have in our bodies,” according to Matthias Hebrok, senior author of a study published last week in the journal Nature Cell Biology.
“This is a critical step toward our goal of creating cells that could be transplanted into patients with diabetes,” Hebrok, director of the UCSF Diabetes Center, said in a statement.
Islets of Langerhans are groupings of cells that contain healthy beta cells, among others. As beta cells develop, they have to separate physically from the pancreas to form these islets.
The team artificially separated the pancreatic stem cells and regrouped them into these islet clusters. When they did this, the cells matured rapidly and become responsive to blood sugar. In fact, the islet clusters developed in ways “never before seen” in a lab. After producing these mature cells, the team transplanted them into mice. Within days, the cells were producing insulin similar to the islets in the mice. While the study has been successful in mice, it still needs to go through more rigorous testing to see if it would work for humans as well. But the research is up-and-coming. “We can now generate insulin-producing cells that look and act a lot like the pancreatic beta cells you and I have in our bodies. This is a critical step towards our goal of creating cells that could be transplanted into patients with diabetes,” He said.
“We’re finally able to move forward on several different fronts that were previously closed to us,” he added. “The possibilities seem endless.”
Basic research keeps elucidating new aspects of beta cells; there seem to be several subtypes, so the gold standard for duplicating the cells is not entirely clear. Today, however, there is “a handful of groups in the world that can generate a cell that looks like a beta cell,” says Hebrok, who currently acts as scientific advisor to Semma and Sigilon, and has previously advised ViaCyte. “Certainly, companies have convinced themselves that what they have achieved is good enough to go into patients.”
The stem cell reprogramming methods that the three companies use to prompt cell differentiation create a mixture of islet cells. Beta cells sit in pancreatic islets of Langerhans alongside other types of endocrine cells. Alpha cells, for example, churn out glucagon, a hormone that stimulates the conversion of glycogen into glucose in the liver and raises blood sugar. Although the companies agree on the positive potential of islet cell mixtures, they take different approaches to developing and differentiating their cells. Semma, which was launched in 2014 to commercialize the Harvard group’s work and counts Novartis among its backers, describes its cells as fully mature, meaning that they are wholly differentiated into beta or other cells before transplantation. “Our cells are virtually indistinguishable from the ones you would isolate from donors,” says Semma chief executive officer BastianoSanna
To get around the donor problem, researchers, including the team at UCSF has been working on nudging stem cells into becoming fully-functional pancreatic beta cells for the last few years. Still, there have been some issues in getting them all the way there.
“The cells we and others were producing were getting stuck at an immature stage where they weren’t able to respond adequately to blood glucose and secrete insulin properly,” Hebrok said.
“It has been a major bottleneck for the field.”
“We’re finally able to move forward on a number of different fronts that were previously closed to us,” Hebrok added. “The possibilities seem endless.”
Regardless of starting cell type, the companies say they are ready to churn out their cells in large numbers. Semma, for example, can make more islet cells in a month than can be isolated from donors in a year in the United States, Sanna says, and the company’s “pristine” cells should perform better than donor islets, which are battered by the aggressive techniques required for their isolation.
As these products, some of which have already entered clinical trials, move toward commercialization, regulatory agencies such as the US Food and Drug Administration (FDA) and the European Medicines Agency have expressed concern about the plasticity of the reprogrammed cells. All three firms subject their cells to rigorous safety testing to ensure that they don’t turn tumorigenic. Before successful trials, companies won’t know the dose of beta cells required for a functional cure, or how long such “cures” will last before needing to be boosted. There’ll be commercial challenges, too: while the companies are investing heavily to develop suitable industrial processes, all acknowledge that no organization has yet manufactured cell therapies in commercial volumes.
Nevertheless, there’s growing confidence throughout the field that these problems will be solved, and soon. “We have the islet cells now,” says Alice Tomei, a biomedical engineer at the University of Miami who directs DRI’s Islet Immuno-engineering Laboratory.
“These stem cell companies are working hard to try to get FDA clearance on the cells.”
Protecting stem cell therapies from the immune system
Whatever the type of cell being used, another major challenge is delivering cells to the patient in a package that guards against immune attack while keeping cells fully functional. Companies are pursuing two main strategies:
- Microencapsulation, where cells are immobilized individually or as small clusters, in tiny blobs of a biocompatible gel.
- Macroencapsulation, in which greater numbers of cells are put into a much larger, implantable device.
ViaCyte, which recently partnered with Johnson & Johnson, launched its first clinical trial in 2014. The trial involved a micro-encapsulation approach that packaged up the company’s partially differentiated, ESC-derived cells into a flat device called the PEC-Encapsulation. About the size of a Band-Aid, the device is implanted under the skin, where the body forms blood vessels around it. “It has a semipermeable membrane that allows the free flow of oxygen, nutrients, and glucose,” says ViaCyte’s chief executive officer, Paul Laikind. “And even proteins like insulin and glucagon can move back and forth across that membrane, but cells cannot.”
The trial showed that the device was safe, well-tolerated, and protected from the adaptive immune system—and that some cells differentiated into working islet cells. But most cells didn’t engraft effectively because a “foreign body response,” a variant of wound healing, clogged the PEC-Encap’s membrane and prevented vascularization. ViaCyte stopped the trial and partnered with W. L. Gore & Associates, the maker of Gore-Tex, to engineer a new membrane. “With this new membrane,” says Laikind, “we’re not eliminating that foreign body response, but we’re overcoming it in such a way that allows vascularization to take place.” The company expects to resume the trial in the second half of this year, provided it receives the green light from the FDA.
Semma is also developing macro¬-encapsulation methods, including a very thin device that in prototype form is about the size of a silver dollar coin. The device is “deceptively simple, but it allows us to put [in] a fully curative dose of islets,” Sanna says.
Semma is also investigating microencapsulation alternatives. At the same time, the company is advancing toward clinical trials using established transplantation techniques to administer donated cadaver cells to high-risk patients who find it particularly difficult to control their blood glucose levels. These cells are infused via the portal vein into the liver, and patients take immunosuppressive drugs to prevent rejection.
Sigilon is working on its microencapsulation technology. Launched in 2016 on the back of work by the labs of Robert Langer and Daniel Anderson at MIT, the company has created 1.5-millimeter gel-based spheres that can hold between 5,000 and 30,000 cells (Nat Med, 22:306–11, 2016). Each sphere is like a balloon, with the outside chemically modified to provide immune-protection, says Sigilon chief executive officer Rogerio Vivaldi. “The inside of the balloon is full of a gel that creates almost a kind of a matrix net where the cells reside.”
In 2018, shortly after partnering with Eli Lilly, Sigilon and collaborators published research showing that islet cells that were encapsulated in gel spheres and transplanted into macaques remained functional for four months. The company has not disclosed a time frame for a type 1 diabetes trial “but we’re moving pretty quickly,” says chief scientific officer David Moller.
Conclusion
To conclude, all three firms hope to extend their work to treat some of the 400 million people worldwide with type 2 diabetes, many of them eventually benefit from insulin injections. The recent endorsements from big Pharmaceutical underline the real progress in beta-cell transplants, says Aaron Kowalski, a molecular geneticist and chief executive officer at JDRF, a foundation based in New York that has funded research at ViaCyte and academic labs whose work has been tapped by Semma and Sigilon. “These companies all realize that if they don’t do it, somebody else will. It’s hard to predict exactly when, but somebody is going to make this work.”
- Published in Corporate News / Blog
Bats Carry Corona Virus. So Why Don’t They Get Sick?
A lot of viruses that has taken a toll on life, the ebola virus in Africa, The Nipah virus of Nipah and the most recent one corona virus that left china running helter skelter all seemed to have originated from bats. During the course of the virus epidemic in Wuhan where it was first detected, some Chinese researchers in Wuhan examined some patients affected in that area and then took samples of the virus.
They did findings on the genetic sequence of the virus with other viruses that were known. The corona virus surprisingly had a 96% match with the horseshoe bats that are dominant in the southwest of china. The research findings were then published in a study on February, 2020.
A virologist Vineet Menachery from the university of Texas Medical Branch at Galveston though not affiliated to the study said “They’re too close in terms of their pure genetics to say they’re not related, or that they didn’t have a common ancestor.”
Menachery was a reputable virologist and had done other research works. He contributed to the theory that the spread of the corona virus must have been from these bats to humans. And possibly must have had another animal that served as an intermediary for the spread.
This same thing had happened with other forms of corona viruses as noted in the case of SARS (Severe Acute Respiratory Syndrome) an outbreak that took place in 2002-2003 where civets, a mongoose family member were infected with the bat corona virus and spread as humans bought them for food.
Another case was the MERS (Middle East Respiratory Syndrome) outbreak. This one happened in 2012 and was as a result of infected camels from the virus. People who ate undercooked meat of camels and as well drank the raw milk of camels were all affected.
So why is it that there are so many diseases that are spread from bats?
Its no doubt, bats have a lot of viruses that they carry with them. And these viruses in their variety are spread and manifests its tolls on people. Scientist are not sure why this is the case as confirmed by Kevin Olival, a research vice president as EcoHealth Alliance, a non-profit organization based in the U.S. He went further to say that it may have something to do with the family of the viruses carried by the bats. So you know, there are over 130 different families of viruses that bats do carry around.
And then, most bats and humans do come in contact through several means. The millions of populations of bats are ubiquitous to all the continents apart from in antartica. Rebekah of Colorado State university who researched infectious pathogens said “There’s a lot of viruses we’re finding in bats because there’s a lot of bats out there.”
They move about in multitudes and live in colonies of large populations. Some these members live in caves and share caves and trees where there can be a contact between humans and bats. Hence, these viruses can spread from these bats to humans.
Despite their sizes, bats have relatively long lifespans and can live over 30 years.”So there’s a long time for them to be persistently infected with the virus and shed it into the environment,” Kading says. The mode of mechanisms for these viruses are through urine, saliva and feces of bats. The outbreak of Nivah that happened in Bangladesh was linked to the sap of a date palm gotten from some trees that some bats licked and had infested with their urine.
Reading through all these, it is not absurd to wonder why the bats themselves do not get affected by the viruses they carry.
The answer to that question is based on the fact that the bat is the only flying mammal in the world. Their body metabolism and process quite differ from that of normal mammals too. When bats fly, their heart rates rise to about a thousand beats per minute with a temperature rise of about 100 degrees Fahrenheit. Linfa Wang a student of bat viruses at Duke-NUS Medical School in Singapore says that when these signs manifest in other mammals, they are signals that can trigger death. But this is not the same case for other bats. This is a lifestyle for them, every day.
Their system is also capable of producing molecules that other organisms do not have. The molecules carry out repair functions and prevent cell damage. This makes their system a bit irresistible to infections and also make them recalcitrant to viruses and resilient to diseases such as diabetes, cancer and other health conditions.
This is a prove that the manifestation of viruses in mammals is not always as a result of the virus itself, but as a result of the body’s reaction to the presence of such a virus that makes us ill by triggering other chain reactions, as Wang explains.
Olival at EcoHealth explains that these bats have coevolved with these viruses and it is not totally their fault that we humans are infected and affected by these viruses. The actual problem is when the viruses move from their species to other species of mammals which is also fostered by human activity.
Naturally, it would be hard for most animals and mammals to cross paths. But Olivial says that the presence of some activities and availability of exchange platforms made available by humans can allow such interaction to occur. She gave an example using wildlife markets like the one in Wuhan, where a bat could be mixed up with a civet. Who later on come in contact with humans – eg. Butchers who do not observe proper hygiene and protection from animal blood.
“The way that we’re coming into contact with these animals, hunting, selling, and trading them is to a scale that really we haven’t seen before,” he says.
Investigative teams did some in-depth search and they discovered some traces of this virus in 22 stalls and in a garbage truck that was found at Huanan Seafood Market right there in Wuhan, a place known for booming trade for live animals. This discovery led to shutting down the market as it was tied to majority of the cases.
The intermediary animals to this viruses are still a mystery, but it is clear that some of these animals are prone to interact more with humans. This is why when they are infected, the likelihood for human infection is widened. These other infected animals can sneeze, urinate, be cooked as food or even owned as pets.
Bats are not just vectors for viruses, they play an important role in balancing the eco-system. They feed on insects and fruits and are active agents of pollination. In fact, Wang believes that since these bats have successfully coevolved with these viruses, there is every possibility that they can be the agents that can lead to the cure and provision of therapies for these viruses.
- Published in Corporate News / Blog
Bats Carry Corona Virus. So Why Don't They Get Sick?
A lot of viruses that has taken a toll on life, the ebola virus in Africa, The Nipah virus of Nipah and the most recent one corona virus that left china running helter skelter all seemed to have originated from bats. During the course of the virus epidemic in Wuhan where it was first detected, some Chinese researchers in Wuhan examined some patients affected in that area and then took samples of the virus.
They did findings on the genetic sequence of the virus with other viruses that were known. The corona virus surprisingly had a 96% match with the horseshoe bats that are dominant in the southwest of china. The research findings were then published in a study on February, 2020.
A virologist Vineet Menachery from the university of Texas Medical Branch at Galveston though not affiliated to the study said “They’re too close in terms of their pure genetics to say they’re not related, or that they didn’t have a common ancestor.”
Menachery was a reputable virologist and had done other research works. He contributed to the theory that the spread of the corona virus must have been from these bats to humans. And possibly must have had another animal that served as an intermediary for the spread.
This same thing had happened with other forms of corona viruses as noted in the case of SARS (Severe Acute Respiratory Syndrome) an outbreak that took place in 2002-2003 where civets, a mongoose family member were infected with the bat corona virus and spread as humans bought them for food.
Another case was the MERS (Middle East Respiratory Syndrome) outbreak. This one happened in 2012 and was as a result of infected camels from the virus. People who ate undercooked meat of camels and as well drank the raw milk of camels were all affected.
So why is it that there are so many diseases that are spread from bats?
Its no doubt, bats have a lot of viruses that they carry with them. And these viruses in their variety are spread and manifests its tolls on people. Scientist are not sure why this is the case as confirmed by Kevin Olival, a research vice president as EcoHealth Alliance, a non-profit organization based in the U.S. He went further to say that it may have something to do with the family of the viruses carried by the bats. So you know, there are over 130 different families of viruses that bats do carry around.
And then, most bats and humans do come in contact through several means. The millions of populations of bats are ubiquitous to all the continents apart from in antartica. Rebekah of Colorado State university who researched infectious pathogens said “There’s a lot of viruses we’re finding in bats because there’s a lot of bats out there.”
They move about in multitudes and live in colonies of large populations. Some these members live in caves and share caves and trees where there can be a contact between humans and bats. Hence, these viruses can spread from these bats to humans.
Despite their sizes, bats have relatively long lifespans and can live over 30 years.”So there’s a long time for them to be persistently infected with the virus and shed it into the environment,” Kading says. The mode of mechanisms for these viruses are through urine, saliva and feces of bats. The outbreak of Nivah that happened in Bangladesh was linked to the sap of a date palm gotten from some trees that some bats licked and had infested with their urine.
Reading through all these, it is not absurd to wonder why the bats themselves do not get affected by the viruses they carry.
The answer to that question is based on the fact that the bat is the only flying mammal in the world. Their body metabolism and process quite differ from that of normal mammals too. When bats fly, their heart rates rise to about a thousand beats per minute with a temperature rise of about 100 degrees Fahrenheit. Linfa Wang a student of bat viruses at Duke-NUS Medical School in Singapore says that when these signs manifest in other mammals, they are signals that can trigger death. But this is not the same case for other bats. This is a lifestyle for them, every day.
Their system is also capable of producing molecules that other organisms do not have. The molecules carry out repair functions and prevent cell damage. This makes their system a bit irresistible to infections and also make them recalcitrant to viruses and resilient to diseases such as diabetes, cancer and other health conditions.
This is a prove that the manifestation of viruses in mammals is not always as a result of the virus itself, but as a result of the body’s reaction to the presence of such a virus that makes us ill by triggering other chain reactions, as Wang explains.
Olival at EcoHealth explains that these bats have coevolved with these viruses and it is not totally their fault that we humans are infected and affected by these viruses. The actual problem is when the viruses move from their species to other species of mammals which is also fostered by human activity.
Naturally, it would be hard for most animals and mammals to cross paths. But Olivial says that the presence of some activities and availability of exchange platforms made available by humans can allow such interaction to occur. She gave an example using wildlife markets like the one in Wuhan, where a bat could be mixed up with a civet. Who later on come in contact with humans – eg. Butchers who do not observe proper hygiene and protection from animal blood.
“The way that we’re coming into contact with these animals, hunting, selling, and trading them is to a scale that really we haven’t seen before,” he says.
Investigative teams did some in-depth search and they discovered some traces of this virus in 22 stalls and in a garbage truck that was found at Huanan Seafood Market right there in Wuhan, a place known for booming trade for live animals. This discovery led to shutting down the market as it was tied to majority of the cases.
The intermediary animals to this viruses are still a mystery, but it is clear that some of these animals are prone to interact more with humans. This is why when they are infected, the likelihood for human infection is widened. These other infected animals can sneeze, urinate, be cooked as food or even owned as pets.
Bats are not just vectors for viruses, they play an important role in balancing the eco-system. They feed on insects and fruits and are active agents of pollination. In fact, Wang believes that since these bats have successfully coevolved with these viruses, there is every possibility that they can be the agents that can lead to the cure and provision of therapies for these viruses.
- Published in Corporate News / Blog
A New Medical Device, in the Management of Complex Wounds
- Published in Corporate News / Blog
A New Medical Device, in the Management of Complex Wounds
Because of the complex nature of wound healing process, an injury on the skin can pose several challenges and are likely pose complications especially when they are acute. They can as well deteriorate from acute to chronic conditions which will require external intervention best understood by a specialist physician to get the area affected by the wound under normalcy.
The complexity of wound healing and research remains an ocean of knowledge that is continuously researched intensely to uncover depths of wound healing techniques and interventions. Hence, this report contains an introduction and details to the use of a new medical innovation called Gcells used primarily for the management of wounds in their different etiology.
In a case where the process of wound healing seemed difficult, Gcells proved great effects an attribute to their design and working protocol. Gcells are conditioned to work with an enriched suspension of progenitor cells that can efficiently aid tissue repair process. In this case report, two subjects were used as donors and acceptors of these micro-grafts.
Introduction
The skin is an outer layer of the body, offering protection to the underlying layers. A wound breaks this layer and inhibits the various functions as well as expose or also break the underlying layer of tissues. Repair processes are inherent and part of homeostatic processes of the body to try to restore the skin back to its normalcy in structure and in function.
The basics for the skins repair mechanism is represented by a cloth and an inflammation where vessels dilate and monocytes activate leading to breakdown of necrotic tissues. This basic process can be inhibited or delayed by a number of varying factors that lead to deteriorative transformation of acute wounds to chronic forms. But if there is no alteration in the repair process, Mesenchymal cells kickstart proliferative process and begin to repair and restructure the affected tissues starting from the base. At the same time epithelial tissues begin to grow around the wound leading to a final step of the healing process. In this final stage, remodeling of the skin structure is primary and then maturation of a scar.
These processes are efficient best in certain conditions which if affected by factors such as cardiovascular ailment, diabetes, bacterial or any other genre of infection, can inhibit these processes.
Hence, it is necessary to understand in details these processes if there is going to be development or innovation for effective healing processes. Just as stated above, during the proliferative phase of wound healing, Mesenchymal cells are the key role players. Their structure includes a Mesenchymal stem cell (MSCs), multi potent in nature and offer supportive, therapeutic and trophic functions. They are also able to release viable trophic, anti-inflammatory cytokines and anti-apoptotic molecules that offer protection during the repair of wounded skin. MSCs also possess subpopulations that are stem-like nature commonly referred to as “side population” (SP) they have been found out to be enriched in over 1000-fold of progenitor cells and multipotent stem cells and as well exist in tissues and tumors. SP exists in a variety of organs and tissues, after an original discovery to be prominent in the bone marrow of a mouse. The organs with SP include the lung, liver, brain, mammary gland and in skeletal muscles.
In other discoveries, it was discovered that they probably may also be isolated in other tissues of the body. This discovery was in an in vitro and in vivo experiment when Dental Pulp Stem Cells (DPSCs) showed capability to differentiate into osteoblasts and built a woven bone by forming an Extracellular Matrix (ECM) secreted by the osteoblasts. The experiment drew results on the both the quality and quantity of the matrix formed by the DPSCs in the in vivo and in vitro experiment using Stem cells and accompanying biomaterials.
Thus proved that dental pulp holds potentialities of therapeutic strength and a rich source of progenitor/autologous cells that can be used to aid healing processes even applicable to regeneration of craniofacial bones.
This is the evidence that supports the working principle of Gcells innovation. Gcell successfully separates this side population with a size of 50 micron. At this cell population, they can form autologous micro-grafts and can either be used alone or alongside biomaterials prepared in a biocomplex ready for use when necessary.
In this case report, two subjects were used as donors and acceptors of these micro-grafts for enhanced healing of complex wounds through autologous micro-grafts using the Gcell.
Clinical case 1
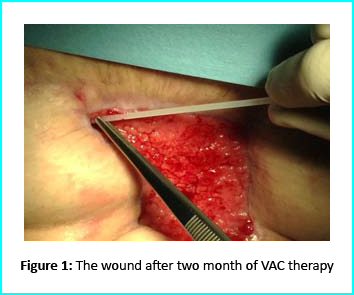
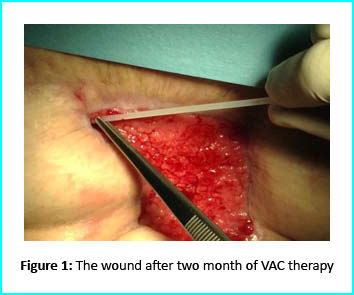
The first case involves a woman at age 50 who does not have any diseases or disorders. She underwent a laparoscopic gastric bypass surgery and was doing well considering parameters of weightloss. Two years later she moved in for abdominoplasty bariatric. Later on, post complications showed preeminence of necrosis which was discovered after first medical examination were about 150 to 200cm2 at the end of the flaps. An initial necrosectomy showed an intense loss of tissue and we furthered to place the wound on VAC therapy and the patient in active participation of this therapy for one week then at home as an outpatient.
As an outpatient, there was improved and progressive wound cleansing while granulated tissues around the base area were cleared.
The VAC therapy after 2 months still left the margins of the wound deteriorated and surrounding areas not in axis with skin surface.
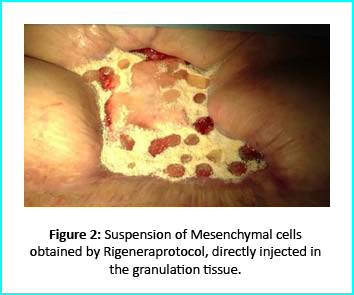
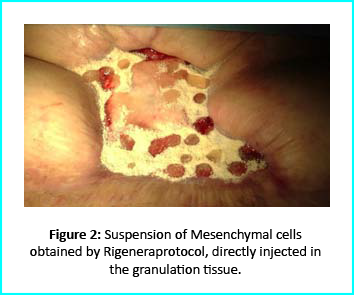
The Gcell protocol kickstarted after consent from the outpatient. We started by collecting a 3 cm2 skin sample from the patient for the purpose of obtaining the cell suspension needed to be injected to the granulation tissue (figure2).
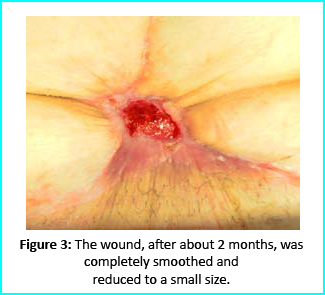
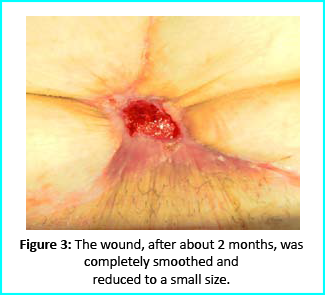
We followed up with conventional wound treatment as in cleaning and replacement with sterile gauze dabbed with Vaseline. The wound area began to improve in both healing progress and general appearance. In two months, the undermined area disappeared as well as leveled to the axis of the skin surface. 2 months later, the wound reduced to a very little scar that is mild and smoothed compared to the initial condition. (figure 3).
A man who suffered liver cirrhosis, hiatal hernia and diabetic as well at the age of 78. Complex surgery was carried out and distal esophagectomy was performed. But hiatal hernia was not decreased into the abdomen, so he was booked up for corrective surgery. During the intervention the adhesions correlated to the previous abdominal operation and led to opening the colon for resection. Some postoperative complications by the appearance of entero-cutaneous fistulas, related to a colonic anastomosis dehiscence. A second intervention was inevitable hence a ileostomy protection and repackaging of colonic anastomosis. We closed the laparotomy using a biological prosthesis. But we met further complications from ascetic failure that needed intensive care hepatology.
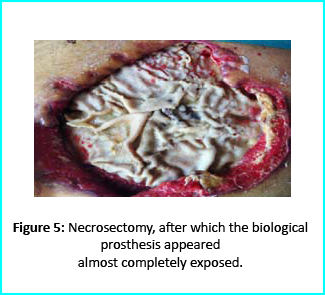
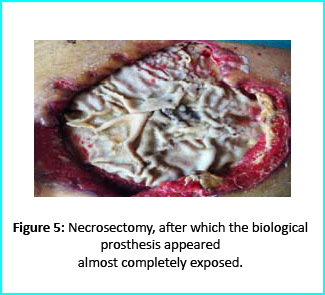
Patient’s condition that included poor liver synthesis had its toll on the healing of the surgical wound. Just as the first case, necrotic tissues grew around the biological prosthesis. We conducted necrosectomy and the biological prosthesis was left half exposed. (Figure 5).
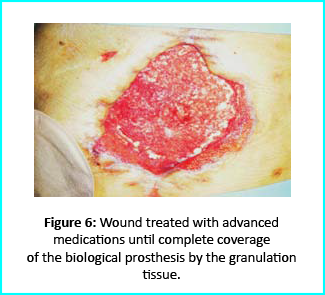
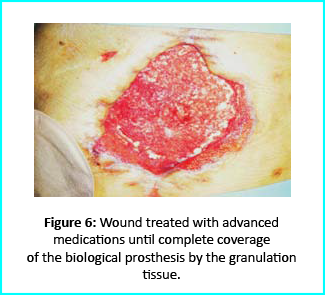
Further treatment of the wound using advanced medication helped cover the biological prosthesis with granulation tissue (figure 6).
Plastic surgeons conducted evaluations on the patient and the choice to do a rotation flap did not seem so appropriate. VAC therapy was used on the wound for about 15 days even though the device wasn’t efficient enough to maintain supposed suction in the presence of ileostomy. We proceeded to treat the patient further with Gcell protocol when wound dimension progressed to about 250cm2. The tissue granulation was of right margin near the ileostomy improved even though it appeared to be undermined. In summary, Gcell protocol has proved a great level of efficiency in healing and restoration of damaged tissues. This progress is certain to open way for employment in the clinical practice that involves the treatment and management of acute and chronic wounds and in any other field of medicine that will inevitably need an instrument to repair lesion on tissues.
Discussion and conclusion
We made it clear earlier in this document about the efficiency of Gcell protocol in its aid to wound healing especially for wounds that are likely to develop from acute to chronic conditions. The working principle for the Gcell used to obtain the viable progenitor cells used for the micrografts relies on one individual as both the donor and the acceptor. This will help to reduce complications that are related to implants or injected micrografts that are non-autologous. Gcell is flexible and can be used both during in operating rooms as well as in ambulatories. This innovation is vastly spreading and currently used in the fields of oral-maxillo-facial field proven by recent studies even though a greater area of its application widespread and acceptable in plastic surgery, dermatology and orthopedics.
Conclusion of this report brings to clarity in demonstration, an efficient, useful and low-risk innovation in the field of medicine, useful for areas in wound management and healing. However, the viability of the Gcell product still needs to be texted on subjects with different conditions and perspectives. But we assure that this device will prove to be a better therapeutic approach in the field of medicine in improving healing of complex wounds. This confidence lies in the excellent features and working principles of this device in obtaining cell suspension, flexibility, facility for procedure and more importantly, the cost. This will help reduce the use of exorbitantly prices devices for advanced medication. In summary, apart from introducing an efficient innovation in the medicine. Gcell has the potentialities to offer employment on clinical procedures that will help aid in the management of wounds no matter how the case may be.
- Published in Corporate News / Blog
Knee replacement alternatives
One of the amputating surgeries in the field of medicine is a knee replacement. It involves removing the knee joint and replacing it with a modified prosthesis. However, several modifications of this surgery have been introduced into the high-powered world of surgery, including several alternatives for knee replacement. In this article, we are going to review the several modifications and knee replacement alternatives therein.
What is Knee Replacement?
Knee replacement, also known as knee arthroplasty, is a surgical procedure that involves the amputation or cutting out of a knee joint, the bones reams by a doctor, especially due to accidents or joint ailments such as arthritis. When the bone is removed, it is then replaced with a prosthetic device. Knee replacement can be partial, where selected or affected parts of the joints can be removed, such as the medial, lateral, and anterior compartments can also be removed and replaced with a modified prosthetic.
Why Should You Be looking for a Knee Replacement Alternatives?
Due to the dynamics of the human body, what works for the goose may not necessarily work for the gander. Certain post-symptoms of a knee replacement can be unbearable for most patients.
Pain After Knee Replacement.
Due to pain in the knee joint, a lot of patients embark on this old-time surgery to help reduce the pain they feel around their knee. But it is worthy of knowing that a substantial number of these patients still continue to feel pain after this audacious surgery. In a survey done by the government, 40% of patients that underwent knee replacement experienced miniature pain for over 3-4 years, while another 44% still felt some 3-5/10 degree of pain in 3-4 years. So, it is not worthy of looking in the direction of knee replacement alternatives in order to solve knee pain.
Knee Replacement Risks.
There is a risk in everything that we do, business, taking a walk, climbing a hill. Same way, certain risks exist in knee replacement which are:
- Patients become more susceptible to heart attack and stroke immediately after knee replacement surgery.
- Increased levels of metals in the blood.
- Allergic reactions to the prosthetic material.
- Possibility of infection.
- Reduced activity of the patient as they thrive to become accustomed to the new prosthesis.
Even though social media and digital marketers paint a vivid picture of beautiful seniors riding a bike, continuing in their daily activities and hobbies, but this may not be true for everyone; in a study conducted by the government, there was seldom activity by patients after knee replacement surgery. Another study showed that patients who weren’t running before a knee replacement surgery couldn’t run after the surgery. But there are always two ways to everything; some other patients also showed an increase in physical activity after their surgery.
What are Knee Replacement Alternatives?
Steroid Injections
Steroids are made up of corticosteroids and cortisone. These corticosteroids carry out an anti-inflammatory function to prevent swelling around the knee regions as well as help reduce pain. But they do have a side effect; they destroy cartilage and may not be efficient as they are thought to be. If you are considering this knee replacement alternative, you probably should bear in mind that they do not offer long term remedies. Steroid injections are viable for knee replacement needs caused by arthritis but may proffer short-termed solutions.
Viscosupplementation
Viscosupplementation is also another knee replacement alternative. They are in the form of gels for the knee, also knowns as hyaluronic acid varying across different brands in the market, likes of SynVisc, OrthoVisc, Supartz, and Euflexxa. They are administered to the patient, but a quick question one would ask is if really the shots help. The variations of results all over the web show support both sides of the notion. But one peculiarity of these results is that none says that they are hurtful or damaging as the steroid injections rather that they give a better solution to knee joint arthritis patients. In my own experience, these injections are efficient only when administered a few times, after which they begin to diminish in effects. The first dose may offer relief for some time, but a dose a far-reaching as the sixth dose may not offer any remedial effect at all.
Knee Nerve Ablation
Knee Nerve ablation is another breakthrough in the surgical world. Knee Nerve Ablation involves the use of technology to carry out a process where the specialist probes the nerves around the joint and passes electrical energy that is used to ablate (destroy) them. The work of these nerves is to relay signals from that region of the knee to the brain. So this technique deadens these nerves, and as such, you don’t feel any pain till those nerves grow back. The research on this type of knee replacement alternative is only a handful. Hence, they cannot conclude on the long term results since most of the studies on this new breakthrough are in their early stages.
Orthobiologics
Orthobiologics incorporation around the knee regions helps to enhance the healing of the knew joint or reduce the consequent degradation of orthopedic tissues. Orthobiologics are also knee replacement alternatives and can be gotten from the patient as autologous or a donor as allogeneic. The two primary derivations of orthobiologics are the PRP and the BMC short for Bone Marrow Concentrate. Another derivation that is commonly used is derived from natal tissues as in amniotic or umbilical cord. Just as the nerve ablation, the research on this type of knee replacement is at its early stages.
Platelet Rich Plasma (PRP)
We mentioned PRP earlier while discussing orthobiologics. PRP’s stand for Platelet-rich plasma that can be gotten from the patient. They contain healing factors that allow them to foster cartilage repair as well as reduce inflammation and balance the chemical dynamics of the knee. A lot of studies support the efficiency of PRP as knee replacement alternatives but may not offer much help when the arthritis is severe.
PKA (Percutaneous Knee Arthroplasty)
PKA (Percutaneous Knee Arthroplasty) comes in handy for severe cases of arthritic pain. This procedure involves the injection of rich bone marrow concentrates gotten from the patient or from a donor into the lax ligaments or other affected areas such as damaged meniscus tissues and tendons. This procedure is intricate and uses an ultrasound and fluoroscopy guides as compared to other quick knee shot techniques. Research proves that this method works pretty well, even in extreme cases of knee arthritis. This procedure also produces a lasting effect for about 2-7 years before the need for repetition.
Here you go!! Knee replacement alternatives. You sure would want to consider some of the alternatives; likes of PKA, PRP, and Bone Marrow concentrates that proffers a long-lasting solution.
- Published in Corporate News / Blog
ISSCA Conference at University of Miami Attracts Regenerative Medicine Experts and Physicians from across the Globe
The regenerative medicine symposium was held on October 24-27 with nearly 200 attendees and over 20 expert speakers
MIAMI LAKES, Florida—The International Society for Stem Cell Application (ISSCA), in collaboration with SISDET, held a highly successful three-day medical conference on the University of Miami campus on October 24-26. The conference featured a host of international experts in regenerative medicine and introduced new standards in regenerative medicine protocols to those in attendance. The Miami conference is part of the ISSCA’s growing commitment to increasing the awareness and practice of regenerative medicine across the globe in an effort to help alleviate suffering for those diagnosed with degenerative diseases.
Around 200 physicians, scientists, and researchers interested in regenerative medicine traveled to the University of Miami campus for the event. The conference focused on providing attendees with information on today’s most successful stem cells treatment protocols and the latest advances in regenerative medicine. Attendees heard from more than 20 expert speakers within the stem cells field, with lecturers from Europe, the US, and Latin America on the conference agenda.
“This three-day event included recognized keynote speakers, as well as aspiring young physicians discussing the latest advances in stem cell biology in an informal and collaborative setting,” said Benito Novas, Vice President of Public Relations for ISSCA. “Our goal with all of our events is to strengthen the cooperative and dynamic spirit in this research area. We would also like to thank the University of Miami for hosting this event, as it was a great honor partnering with such a prestigious university.”
ISSCA is a global leader in stem cells research, applications, and education, partnering with major global institutions and locations worldwide to host its independent medical congresses. To learn more about the ISSCA and its all of its past and upcoming events, visit http://www.issca.us
- Published in Corporate News / Blog
Phase I/II Study of Safety and Preliminary Efficacy of Intravenous Allogeneic Mesenchymal Stem Cells in Chronic Stroke
Phase I/II Study of Safety and Preliminary Efficacy of Intravenous Allogeneic Mesenchymal Stem Cells in Chronic Stroke
Michael L. Levy, MD, PhD; John R. Crawford, MD; Nabil Dib, MD; Lev Verkh, PhD; Nikolai Tankovich, MD, PhD; Steven C. Cramer, MD
Background and Purpose—Stroke is a leading cause of long-term disability. Limited treatment options exist for patients with chronic stroke and substantial functional de?cits. The current study examined safety and preliminary ef?cacy estimates of intravenous allogeneic mesenchymal stem cells in this population.
Methods—Entry criteria included ischemic stroke >6 months prior and substantial impairment (National Institutes of Health Stroke Scale score =6) and disability. Enrollees received a single intravenous dose of allogeneic ischemia-tolerant mesenchymal stem cells. Phase 1 used a dose-escalation design (3 tiers, n=5 each). Phase 2 was an expanded safety cohort. The primary end point was safety over 1-year. Secondary end points examined behavioral change.
Results—In phase 1 (n=15), each dose (0.5, 1.0, and 1.5 million cells/kg body weight) was found safe, so phase 2 subjects (n=21) received 1.5 million cells/kg. At baseline, subjects (n=36) averaged 4.2±4.6 years poststroke, age 61.1±10.8 years, National Institutes of Health Stroke Scale score 8 (6.5–10), and Barthel Index 65±29. Two were lost to follow-up, one was withdrawn and 2 died (unrelated to study treatment). Of 15 serious adverse events, none was possibly or probably related to study treatment. Two mild adverse events were possibly related to study treatment, a urinary tract infection and intravenous site irritation. Treatment was safe based on serial exams, electrocardiograms, laboratory tests, and computed tomography scans of chest/abdomen/pelvis. All behavioral end points showed signi?cant gains over the 12-months of follow-up. For example, Barthel Index scores increased by 6.8±11.4 points (mean±SD) at 6-months (P=0.002) and by 10.8±15.5 points at 12-months (P<0.001) post-infusion; the proportion of patients achieving excellent functional outcome (Barthel score =95) increased from 11.4% at baseline to 27.3% at 6-months and to 35.5% at 12-months.
Conclusions—Intravenous transfusion of allogeneic ischemia-tolerant mesenchymal stem cell in patients with chronic stroke and substantial functional de?cits was safe and suggested behavioral gains. These data support proceeding to a randomized, placebo-controlled study of this therapy in this population.
Clinical Trial Registration—URL: http://www.clinicaltrials.gov. Unique identi?er: NCT01297413. (Stroke. 2019;50:00-00. DOI: 10.1161/STROKEAHA.119.026318.)
Key Words: abdomen ? brain ischemia ? neuroprotection ? pelvis ? reperfusion
Stroke is perennially among the leading causes of human disability1 and the leading neurological cause of lost dis-ability-adjusted life years.2 The mean survival after stroke is 6 to 7 years, and indeed more than 85% of patients live past the ?rst year poststroke,3 many with years of enduring disability. Many restorative therapies are under study to improve outcomes after stroke.4 Restorative therapies aim to improve patient outcomes by promoting the neural processes under-lying behavioral recovery,5 and are distinguished from acute therapies, such as reperfusion or neuroprotection, that aim to reduce initial injury. As such, restorative therapies often have a time window measured in days-months, or in some cases6–9 in years.
Mesenchymal stem cells (MSC), also known as mes-enchymal stromal cells, are among the leading restorative therapy candidates. Substantial preclinical data support the safety and ef?cacy of MSC as a restorative therapy to improve outcomes after stroke. For example, a meta-analysis reported that 44 of 46 preclinical stroke studies found MSC to be supe-rior to placebo,10 with effect sizes >1.0.
Initial human studies of MSC (or MSC-like cells) after stroke focused on autologous cell therapies,11–13 whereby bone marrow is taken from each patient to produce his/her own MSC batch, and found MSC infusion to be safe. MSC are rela-tively immunoprivileged given their very low levels of human leukocyte antigen molecule expression,14 a fact that opens the door to administration of allogeneic MSC. Allogeneic MSC Table 1. Entry Criteria have been found to be safe without use of concomitant im-munosuppression,15 and can be manufactured in a manner that enables broad clinical application. Studies of allogeneic MSC (or MSC-like cells) poststroke have focused on early time points (administration 24–48 hours poststroke)16 or used an invasive procedure to implant cells intracerebrally.17 Each approach has its relative advantages and disadvantages, and an intravenous method of introducing MSC if comparably ef-?cacious might facilitate widespread implementation and also avoid adverse events attributable to invasive procedures.
The current study was a phase I/II dose-escalation trial that examined effects of a single intravenous infusion of al-logeneic ischemia-tolerant MSC. The target population was patients with chronic ischemic stroke and substantial func-tional de?cits, a group for whom treatment options remain limited. The primary outcome was safety, based on serial measures of behavior, computed tomography (CT) scans, and laboratory testing. Preliminary estimates of treatment ef?cacy were also examined.
Table 1. Entry Criteria
| Inclusion criteria |
| 1. Age =18 y |
| 2. Ischemic stroke =6 mo prior, radiologically confirmed at initial diagnosis and at study enrolment |
| 3. Severe disability resulting from the index stroke, operationally defined as subject confined to a wheelchair or required to have home nursing care or needing assistance with activities of daily living |
| 4. No substantial improvement in neurological or functional status for the 2 mo before study enrolment |
| 5. NIHSS score 6–20 |
| 6. Life expectancy >12 mo |
| 7. Patient receiving standard of care secondary stroke prevention before enrolment |
| 8. Patient or a surrogate able to provide informed consent |
| 9. Reasonable expectation that the patient will receive standard posttreatment care and attend all scheduled study visits |
| 10. Adequate systemic organ function, specifically: Serum aspartate aminotransferase =2.5× upper limit of normal Serum alanine aminotransferase =2.5× upper limit of normal Total serum bilirubin =1.5× upper limit of normal Prothrombin time and partial thromboplastin time =1.25× upper limit of normal in subjects who are not receiving anti-thrombotic therapy Serum albumin =3.0 g/dL Absolute neutrophil count =1500/µL Platelet count =150 000/µL Hemoglobin =9.0 g/dL Serum creatinine =1.5× upper limit of normal Serum amylase or lipase =1.0× upper limit of normal |
| Exclusion criteria |
| 1. History of uncontrolled seizure disorder |
| 2. History of cancer within the past 5 y, with the exception of localized basal or squamous cell carcinoma |
| 3. History of cerebral neoplasm |
| 4. Positive for hepatitis B, C, or HIV |
| 5. Myocardial infarction within 6 months of study entry |
| 6. Presence of any other clinically significant medical or psychiatric condition, or laboratory abnormality, for which study participation would pose a safety risk in the judgment of the Investigator or Sponsor |
| 7. Findings on baseline computed tomography suggestive of subarachnoid or intracerebral hemorrhage within past 12 mo. |
| 8. Participation in another investigational drug or device study in the 3 mo before treatment |
| 9. History within the past year of drug or alcohol abuse |
| 10. Pregnant or lactating, or expectation to become pregnant during the study |
| 11. Allergy to bovine or porcine products |
NIHSS indicates National Institutes of Health Stroke Scale
Methods
Study Design
This was a phase I/II multi-center, open-label study that aimed to evaluate the safety and preliminary ef?cacy of a single intra-venous infusion of marrow-derived allogeneic ischemia-tolerant MSC. Entry criteria appear in Table 1 and in sum describe enroll-ment of adults with radiologically veri?ed chronic stable ischemic stroke and substantial impairment and functional de?cits. Patients were followed for one year after MSC infusion. The study made no restrictions on, and did not provide any forms of, medication or therapy (occupational, physical, or speech) during the follow-up year after infusion. All patients signed consent in accordance with local Institutional Review Board approval. This study was approved by the Food and DrugAdministration and was registered at clinical-trials.gov. The data that support the ?ndings of this study are avail-able from the corresponding author on reasonable request.
The study occurred in 2 parts, with part 1 being a dose-escala-tion study and part 2 being an expanded safety study based on part 1 ?ndings. Part 1 consisted of 3 cohorts (n=5 per cohort) enrolled sequentially in a dose-escalation manner, with subjects receiving one of 3 doses based on body weight, with a maximum dosage of 150 million cells. Cohort 1 received 0.5 million cells/kg of body weight; Cohort 2, 1.0 million cells/kg; and Cohort 3, 1.5 million cells/kg. The dose-escalation plan in part 1 required a review by the Data Safety Monitoring Board once the 5 subjects in Cohort 1 were treated and evaluated through study day 10. If safety was established, Cohort 2 was to proceed at the next highest dose, followed by a similar safety review before escalation to the highest dose in Cohort 3. Part 2 aimed to enroll an additional minimum of 20 subjects at the highest safe dose level determined in part 1. An additional interim review was conducted by the Data Safety Monitoring Board after the ?rst 5 patients were treated in part 2. Detailed stopping rules appear in the online-only Data Supplement (see Stopping Rules and Determination of Maximum Tolerated Dose).
The target dose of 1.5 million cells/kg corresponds to allometric scaling from animal studies. Our meta-analysis of preclinical studies of MSC after experimental ischemic stroke10 identi?ed 9 rodent stud-ies that transfused MSC using the intravenous route in the post-acute period. In each study, MSC provided substantial behavioral gains (effect sizes >1.0), using doses ranging from 3.6 to 12.4×106 MSC/kg body weight (mean dose of 10.1×106 MSC/kg). The approach to al-lometric scaling from animals to humans recommended by the Food and Drug Administration 18 uses a body surface area normalization, which for the mean value in rodents yields a comparable human dose of 1.6×106 MSC/kg.
Cell Manufacturing and Shipping
Manufacturing of MSC was performed at the GMP-compliant fa-cility of the sponsor, Stemedica Cell Technologies, Inc (San Diego, CA). MSC were grown from the bone marrow of a single human donor and are from the same batch used in prior preclinical19,20 and clinical21 studies. Cells were grown under low oxygen (5%) condi-tions. Such ischemia-tolerant MSC have advantages compared with those grown under normoxic conditions, for example, showing higher proliferation rate, expression of stem cell-related genes, production of key cytokines, and migration activity.21,22 Cells were harvested at passage 4 and expressed CD105, CD73, and CD90 surface markers, consistent with the International Society for Cellular Therapy de?-nition.23 Cells were cryopreserved by suspending in Cryostar CS10 freezing medium (BioLife Solutions, Bothell, WA) then stored in the vapor phase of liquid nitrogen. This parent cell bank was then tested for quality control including cell count, viability, appearance, and quantitative polymerase chain reaction for viruses including HIV, Epstein-Barr virus, cytomegalovirus, hepatitis B virus, parvovirus B19, and hepatitis C virus. Cryovials were shipped at =-150° C in a vapor phase liquid nitrogen shipper with temperature monitor.
Infusion of Investigational Product
Each site’s pharmacy prepared MSC for infusion per a study-pro-vided protocol. Cryovials (the number of which was based on the dose to be infused) were thawed and MSC were washed in, and then suspended in, Lactated Ringer’s solution at a concentration of 1×106 cells/mL using one to three 60 mL syringes. The suspension then un-derwent ?nal testing before being released for intravenous infusion, consisting of cell count, endotoxin, Gram stain, and review of appear-ance. Cell count was performed using 0.1% Trypan Blue and a hema-cytometer, which also yielded % cell viability. The minimum percent cell viability was required to be =70% for the cells to be released. A sample was also sent for subsequent sterility testing. After release by the pharmacy, the ?nal formulation was stored at 2° to 8°C and infused within 8 hours of preparation.
MSC Administration
Before MSC infusion, a 0.1 mL aliquot of the ?nal MSC formulation was injected intradermally; any subject showing a positive reaction (eg, wheal with erythema) would not be infused. Cells were admin-istered intravenously via metered-dose syringe pump at 2 mL/min. Patients remained in the inpatient telemetry unit for observation until clinically stable.
Patient Assessments
Patients had frequent monitoring until discharged from the telem-etry unit. After discharge, patients had safety evaluations on day 2, 3, 4, and 10, then again on month, 1, 3, 6, 9, and 12. Adverse events were coded according to the MedDRA adverse event dic-tionary. The relationship that adverse events had to the investiga-tional product was assessed by the site investigator. Patients were followed for one year using tests of behavior, serology, blood chem-istry and cell counts, electrocardiogram, urine, and CT of chest, abdomen, and pelvis. The full schedule of assessments appears in Table SI in the online-only Data Supplement.
Statistics
The primary study end point was safety and tolerability, evaluated in all subjects who received any portion of an infusion, and determined by the incidence/severity of adverse events, clinically signi?cant changes on laboratory and imaging tests, vital signs, and physical plus neurological examinations. Four secondary end points were scored serially to derive preliminary estimates of ef?cacy: National Institutes of Health Stroke Scale, Barthel Index (BI), Mini-Mental Status Exam, and Geriatric Depression Scale. For each, the change from baseline was evaluated using Wilcoxon signed-rank test, with primary analysis of preliminary ef?cacy being change from baseline to 6 months post-infusion, and analysis including all subjects who re-ceived an infusion except for one subject who failed to return after the day 10 visit for all visits (except for month 9 follow-up). For any sub-ject missing 6-month data, 9-month or 12-month data were substituted for this analysis, otherwise missing data were not imputed. Data were analyzed using R statistical software. Given the exploratory nature of this study, sample size was selected as appropriate for detection of any safety concerns in an early phase clinical trial.
Results
Subjects
Of 50 subjects who seemed eligible on prescreening, 36 were enrolled and received treatment from March 14, 2011 to December 15, 2016 (Figure and Table 2). There were 13 subjects enrolled at the University of California, San Diego, 19 subjects at Arizona, and 4 subjects at the University of California, Irvine. Interim safety reviews disclosed no con-cerns, and so 5 subjects received 0.5×106 cells/kg in part 1/ Cohort 1, 5 subjects received 1.0×106 cells/kg in part 1/Cohort 2, 5 subjects received 1.5×106 cells/kg in part 1/Cohort 3, and all 21 subjects in part 2 received 1.5×106 cells/kg. For the 15 subjects in part 1, 12 completed the study, 2 died of unrelated causes (coronary artery disease 6 months post-infusion and sepsis 1 month after infusion), and 1 was lost to follow-up after day 10 (reappearing only for the month 9 follow-up visit). For the 21 subjects in part 2, 19 completed the study, 1 was lost to follow-up after month 6, and 1 was withdrawn by the site PI after month 6 due to treatment with another in-vestigational product. Of the 36 subjects enrolled, the planned dose was delivered within 2 mL (ie, within 2×106 cells) of the target in 26 subjects, whereas in 10 subjects a median of 7.6 (interquartile range, 4.4–10.25) mL (ie, 7.6×106 cells) was not infused as planned, which represented a median of 6.5% (5.3– 9.8) of the intended dose. A total of 179 protocol deviations were reported, mainly related to scheduling study visits or study testing (Table SII in the online-only Data Supplement).
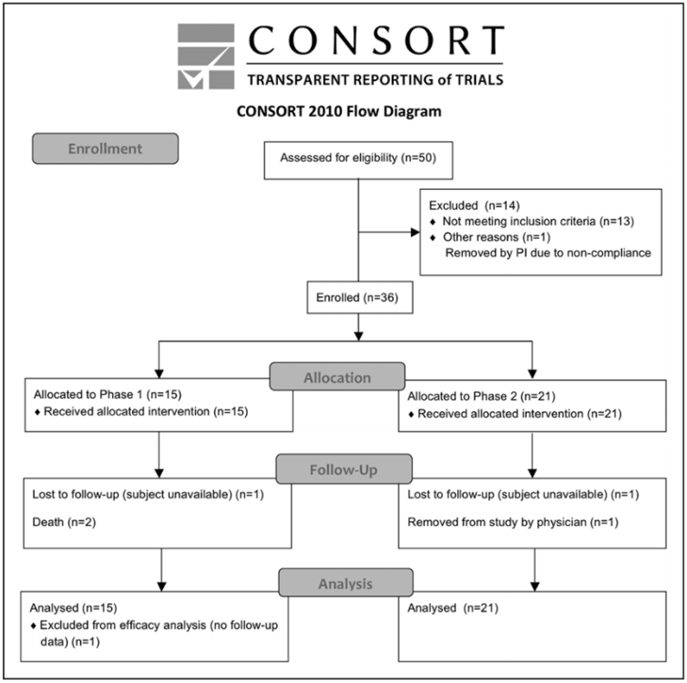
Figure. CONSORT diagram.
Table 2. Baseline Subject Characteristics
| Part1 | Part 2 | Total | |||
| Cohort 1 | Cohort 2 | Cohort 3 | |||
| n | 5 | 5 | 5 | 21 | 36 |
| Sex | |||||
| Male | 5 (100%) | 4 (80%) | 4 (80%) | 14 (66.67%) | 27 (75%) |
| Female | 0 (0%) | 1 (20%) | 1 (20%) | 7 (33.33%) | 9 (25%) |
| Age, y | 50.8:±9.8 [40—62] | 56.8±11.1 [39—69] | 68.8±11.58 [53—84] | 62.8±9.2 [51—83] | 61.1±10.8 [39—84] |
| Race | |||||
| White | 4 (80%) | 3 (60%) | 5 (100%) | 17 (80.95%) | 29 (80.56%) |
| Asian | 0 (0%) | 1 (20%) | 0 (0%) | 0 (0%) | 1 (2.78%) |
| American Indian/Alaskan Native | 0 (0%) | 1 (20%) | 0 (0%) | 0 (0%) | 1 (2.78%) |
| Native Hawaiian/Pacific Islander | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) | 0 (0%) |
| Black | 1 (20%) | 0 (0%) | 0 (0%) | 1 (4.76%) | 2 (5.56%) |
| Other | 0 (0%) | 0 (0%) | 0 (0%) | 3 (14.29%) | 3 (8.33%) |
| Ethnicity | |||||
| Hispanic or Latino | 0 (0%) | 0 (0%) | 0 (0%) | 2 (9.52%) | 2 (5.56%) |
| Non-Hispanic or Non-Latino | 5 (100%) | 5 (100%) | 5 (100%) | 19 (90.48%) | 34 (94.44%) |
| Living situation | |||||
| At home | 5 (100%) | 5 (100%) | 3 (60%) | 19 (90.48%) | 32 (88.89%) |
| In a living facility | 0 (0%) | 0 (0%) | 2 (40%) | 2 (9.52%) | 4 (11.11%) |
| Time from stroke to infusion, y | 1.6±0.9 [0.6—2.9] | 7.7±5.0 [1.1—14.5] | 4.1±2.2 [1.7—7.0] | 4.0±5.0 [0.7—24.8] | 4.2±4.6 [0.6—24.8] |
Values are counts (%) else mean±SD. Values in brackets indicate range.
Safety
A total of 15 serious adverse events were reported.These were wide-ranging in nature, for example, infections, vascular dis-orders, and pain syndromes (for full details, see Table SIII in the online-only Data Supplement). All serious adverse events were deemed unrelated or unlikely related to the investiga-tional product. A total of 109 adverse events were reported, of which 2, both mild, were considered by the site investigator to be possibly related to the investigational product: one uri-nary tract infection and one report of intravenous site irrita-tion. Both adverse events recovered completely.
Study testing disclosed no safety concerns. No subject showed a preinfusion positive reaction to intradermal testing. Serial physical exams and blood testing did not disclose any signi?cant ?ndings. Only one of the serial electrocardiograms was thought to have clinically signi?cant ?ndings, in a subject with moderate intraventricular conduction delay, only at the 1-month follow-up visit. Similarly, across serial CT scans of the chest, abdomen, and pelvis, only one was considered clinically signi?cant, a soft tissue density in the anterior abdominal wall seen at 6-months that was stable when reimaged at 12-months.
Behavioral Effects
Across all subjects, improvements were seen in National Institutes of Health Stroke Scale, BI, Mini-Mental Status Exam, and Geriatric Depression Scale scores at both the 6-month and the 12-month follow-up visits (Table 3). These were statistically signi?cant, generally stable over time, and clinically modest in magnitude. Most ?ndings would sur-vive correction for multiple comparisons. Changes in the BI suggest clinical utility, with a 6.8 point gain by 6-months that grew to a 10.8 point gain by 12-months post-infusion (P<0.001), and with the proportion of patients achieving ex-cellent functional outcome (Barthel score =95) increasing from 11.4% (4/35) at baseline to 9/33 (27.3%) at 6-months to 35.5% (11/31) at 12-months.
Discussion
Stroke is a major cause of human disability. This can be reduced by acute therapies that are introduced in the early hours poststroke to reduce initial injury, and by restorative therapies that are introduced days, months, or years poststroke to promote neural repair. Allogeneic MSC show substantial favorable effects in preclinical studies, including when intro-duced via the intravenous route.10 The current study found a single intravenous infusion of allogeneic MSC to be safe and potentially associated with functional improvement.
The current study is the largest trial of intravenous MSC in patients with chronic stroke and the ?rst to evaluate alloge-neic MSC therapy in this population. It is also the ?rst human stroke study to evaluate MSC grown under hypoxic conditions, which favorably affects cell proliferation, gene expression, cy-tokine production, and migration.21,22 Intravenous infusion of MSCwasfoundtobesafein36patientswhohadchronicstroke with substantial functional de?cits. Across 3 escalating doses, treatment-related adverse events were infrequent, mild, and transient. Serial assessments of exam, laboratory testing, elec-tro cardiogram, and CT scans of chest/abdomen/pelvis disclosed no safety concerns, with limited subject dropout. These results are consistent with the overall excellent safety record that MSC have in clinical trials of human subjects across numerous non-cerebrovascular diagnoses15,24–27 and in stroke trials.11–13,16,17,28,29
Patients with stroke in the chronic stage generally show functional decline; however, enrollees in the current study showed 12 months of continued functional improvement. In general, recovery from stroke-related de?cits shows a bimodal time course. Initially, most stroke survivors show some degree of spontaneous recovery, for example, during the initial months for the motor system.30 Within a year of stroke onset, however, a signi?cant decline in function is commonly seen.31–34 This is signi?cant given that few treat-ment options are available to improve function in patients in the chronic phase of stroke. In the current study, behav-ioral gains were seen, though were modest in magnitude. However, a 2-point improvement in the National Institutes of Health Stroke Scale score (Table 3) in the setting of chronic stroke, if veri?ed in a larger controlled study, might be regarded as important. Also, the mean gain in BI from base-line grew to 10.8 points by 12 month-poststroke (P<0.001), higher than the BI minimal clinically important difference of 9.25 points.35 Furthermore, the proportion of patients with an excellent functional outcome (BI score =95) increased from 11.4% at baseline to 27.3% at 6-months and to 35.5% at 12-months (Table 3). This 12-month period of continued functional improvement is consistent with preclinical studies examining the distribution of systemically administered MSC: intravenous MSC given early after stroke initially localize to lungs then spleen, then increase within the region of brain ischemia,36 and by 30 days poststroke are concen-trated in the peri-infarct region.37 At one year, most surviving MSC are in the peri-infarct region, with very few present in other organs.38 Patients also showed signi?cant improvement in the Mini-Mental Status Exam and Geriatric Depression Scale (Table 3), changes that were largely sustained at 12 months post-infusion, suggesting that MSC have broad effects on brain function. These ?ndings require veri?cation in a larger, controlled study but raise hope that this interven-tion could improve functional status in the chronic stroke setting. Future studies might also incorporate modality-spe-ci?c outcome measures30 to provide more granular assessments of behavioral gains in individual neural systems.
Table 3. Behavioral Change Over Time
| Mini-Mental Status Exam score | n | P Value | |
| Baseline | 24.2±6.0 | 35 | |
| Change to 6 mo | 1.8±2.8 | 32 | <0.001 |
| Change to 12 mo | 1.3±2.7 | 31 | 0.017 |
| NIHSS score | |||
| Baseline | 8 [6.5 to 10] | 35 | |
| Change to 6 mo | -1 [-2.25 to 0] | 32 | <0.001 |
| Change to 12 mo | -2 [-3.5 to -0.5] | 21 | <0.001 |
| Geriatric depression scale score | |||
| Baseline | 5.1±3.5 | 35 | |
| Change to 6 mo | -1.6±3.8 | 32 | 0.015 |
| Change to 12 mo | -1.4±3.8 | 31 | |
| Barthel Index (score) | |||
| Baseline | 65±28.7 | 35 | |
| Change to 6 mo | 6.8±11.4 | 33 | 0.002 |
| Change to 12 mo | 10.8±15.5 | 31 | <0.001 |
| Barthel Index (% =95) | |||
| Proportion at baseline | 4 (11.4%) | 35 | |
| Proportion at 6 mo | 9 (27.3%) | 33 | 0.015 |
| Proportion at 12 mo | 11 (35.5%) | 31 | 0.01 |
Values are mean±SD or median (interquartile range) across all enrollees. Specific data for part 1 and part 2 appear in Table SIV in the online-only Data Supplement. NIHSS indicates National Institutes of Health Stroke Scale.
Meta-analysis of MSC effects in animals with experimental ischemic stroke10 showed large effect sizes that remained sub-stantial after adjusting for potential publication bias and was robust across species, delivery route, time of administration in relation to stroke, and dose. The longest time period when MSC have been introduced poststroke in preclinical studies is 1 month39 or 4 to 6 weeks40 post-infarct. The current ?ndings in patients who were many months poststroke (Table 2) sug-gest the need for bidirectional translation, that is, translation of bedside experience to inform preclinical studies.41,42
There are several strengths to this study. Enrollees had substantial functional de?cits in the chronic stage of stroke, a population that numbers in the millions, for whom treatment options remain limited. The infused cells were allogeneic, an approach made possible by the relatively immunoprivileged nature of MSC,14 which eliminates the need for immuno-suppression15 and which, as compared with autologous cell therapies, enables treatment protocols that can be broadly implemented in the stroke population. A dose-escalation study design was used to evaluate safety. Cell culture was limited to 4 passages, a potential advantage given that higher number of passages (and thus cell divisions) adversely affect MSC features such as proliferation, differentiation, homing, and viability.43–45 Safety was assessed across multiple modalities, including chest/abdomen/pelvis CT and extensive laboratory testing, for a 1-year period.
There are also important weaknesses. As this study was focused on safety, no control group was included, which complicates interpretation of observed behavioral gains (Table 3). Mechanism of action was not studied. Cell thera-pies improving outcomes in the chronic phase likely act via multiple mechanisms that include release of growth factors and anti-in?ammatory effects, and possibly exosomes,46,47 which can be evaluated in subsequent trials. Restorative ther-apies after stroke often provide maximal bene?t when paired with appropriate training,48 but this was not provided in the current safety study.
The current study demonstrated safety of intravenous MSC in patients with chronic stroke who had substantial functional de?cits. Results also suggest functional bene?t, although this requires veri?cation in a controlled study. Together, these ?ndings support further study of intravenous allogeneic MSC in patients with chronic stroke, including evaluation of mech-anism of action.
Sources of Funding
This study was supported by Stemedica Cell Technologies, Inc.
Disclosures
Dr Levy is on the Scienti?c Advisory Board for KOH Robotics. Dr Dib has served as a consultant for J&J Consulting and ISCTR Consulting. Dr Verkh is Chief Regulatory and Clinical Development Of?cer at Stemedica Cell Technologies, Inc. Dr Tankovich is the President and Chief Medical Of?cer for Stemedica Cell Technologies, Inc. Dr Cramer has served as a consultant for Abbvie, Constant Therapeutics, MicroTransponder, Neurolutions, Regenera, SanBio, Stemedica, Biogen, Fuji?lm Toyama Chemical, and TRCare. The other authors report no con?icts.
References
- Feigin VL, Lawes CM, Bennett DA, Anderson CS. Stroke epidemiology: a review of population-based studies of incidence, prevalence, and case-fatality in the late 20th century. Lancet Neurol. 2003;2:43–53.
- Johnston SC, Hauser SL. Neurological disease on the global agenda. Ann Neurol. 2008;64:A11–A12.
- Lloyd-Jones D, Adams RJ, Brown TM, Carnethon M, Dai S, De Simone G, et al. Heart disease and stroke statistics–2010 update: a report from the American Heart Association. Circulation. 2010;121:e46–e215.
- Lin DJ, Finklestein SP, Cramer SC. New directions in treat-ments targeting stroke recovery. Stroke. 2018;49:3107–3114. doi: 10.1161/STROKEAHA.118.021359
- Cramer SC. Repairing the human brain after stroke. II. Restorative thera-pies. Ann Neurol. 2008;63:549–560. doi: 10.1002/ana.21412
- Wolf SL, Winstein CJ, Miller JP, Taub E, Uswatte G, Morris D, et al; EXCITE Investigators. Effect of constraint-induced movement therapy on upper extremity function 3 to 9 months after stroke: the EXCITE randomized clinical trial. JAMA. 2006;296:2095–2104. doi: 10.1001/jama.296.17.2095
- McCabe J, Monkiewicz M, Holcomb J, Pundik S, Daly JJ. Comparison of robotics, functional electrical stimulation, and motor learning methods for treatment of persistent upper extremity dysfunction after stroke: a randomized controlled trial. Arch Phys Med Rehabil. 2015;96:981–990. doi: 10.1016/j.apmr.2014.10.022
- Dodakian L, McKenzie AL, Le V, See J, Pearson-Fuhrhop K, Burke Quinlan E, et al. A home-based telerehabilitation program for patients with stroke. Neurorehabil Neural Repair. 2017;31:923–933. doi: 10.1177/1545968317733818
- Ward NS, Brander F, Kelly K. Intensive upper limb neurorehabilita-tion in chronic stroke: outcomes from the Queen Square programme. J Neurol Neurosurg Psychiatry. 2019;90:498–506. doi: 10.1136/jnnp-2018-319954
- Vu Q, Xie K, Eckert M, Zhao W, Cramer SC. Meta-analysis of preclinical studies of mesenchymal stromal cells for ischemic stroke. Neurology. 2014;82:1277–1286. doi: 10.1212/WNL.0000000000000278
- Bang OY, Lee JS, Lee PH, Lee G. Autologous mesenchymal stem cell transplantation in stroke patients. Ann Neurol. 2005;57:874–882. doi: 10.1002/ana.20501
- Honmou O, Houkin K, Matsunaga T, Niitsu Y, Ishiai S, Onodera R, et al. Intravenous administration of auto serum-expanded autologous mes-enchymal stem cells in stroke. Brain. 2011;134(pt 6):1790–1807. doi: 10.1093/brain/awr063
- Bhasin A, Srivastava MV, Kumaran SS, Mohanty S, Bhatia R, Bose S, et al. Autologous mesenchymal stem cells in chronic stroke. Cerebrovasc Dis Extra. 2011;1:93–104. doi: 10.1159/000333381
- Le Blanc K, Tammik C, Rosendahl K, Zetterberg E, Ringdén O. HLA expression and immunologic properties of differentiated and undifferen-tiated mesenchymal stem cells. Exp Hematol. 2003;31:890–896.
- Lalu MM, McIntyre L, Pugliese C, Fergusson D, Winston BW, Marshall JC, et al; Canadian Critical Care Trials Group. Safety of cell therapy with mesenchymal stromal cells (SafeCell): a systematic re-view and meta-analysis of clinical trials. PLoS One. 2012;7:e47559. doi: 10.1371/journal.pone.0047559
- Hess DC, Wechsler LR, Clark WM, Savitz SI, Ford GA, Chiu D, et al. Safety and ef?cacy of multipotent adult progenitor cells in acute ischaemic stroke (MASTERS): a randomised, double-blind, pla-cebo-controlled, phase 2 trial. Lancet Neurol. 2017;16:360–368. doi: 10.1016/S1474-4422(17)30046-7
- Steinberg GK, Kondziolka D, Wechsler LR, Lunsford LD, Coburn ML, Billigen JB, et al. Clinical outcomes of transplanted modi?ed bone marrow-derived mesenchymal stem cells in stroke: a phase 1/2a study. Stroke. 2016;47:1817–1824. doi: 10.1161/STROKEAHA.116.012995
- Guidance for Industry: Estimating the Maximum Safe Starting Dose in Initial Clinical Trials for Therapeutics in Adult Healthy Volunteers. 2005. www.fda.gov/downloads/Drugs/GuidanceComplianceRegulatory Information/Guidances/ucm078932.pdf.Accessed June 22, 2019.
- Luger D, Lipinski MJ, Westman PC, Glover DK, Dimastromatteo J, Frias JC, et al. Intravenously delivered mesenchymal stem cells: sys-temic anti-in?ammatory effects improve left ventricular dysfunction in acute myocardial infarction and ischemic cardiomyopathy. Circ Res. 2017;120:1598–1613. doi: 10.1161/CIRCRESAHA.117.310599
- Harach T, Jammes F, Muller C, Duthilleul N, Cheatham V, Zufferey V, et al. Administrations of human adult ischemia-tolerant mesenchymal stem cells and factors reduce amyloid beta pathology in a mouse model of Alzheimer ’s disease. Neurobiol Aging. 2017;51:83–96. doi: 10.1016/j.neurobiolaging.2016.11.009
- Butler J, Epstein SE, Greene SJ, Quyyumi AA, Sikora S, Kim RJ, et al. Intravenous allogeneic mesenchymal stem cells for nonischemic car-diomyopathy: safety and ef?cacy results of a phase II-a randomized trial. Circ Res. 2017;120:332–340. doi: 10.1161/CIRCRESAHA. 116.309717
- Vertelov G, Kharazi L, Muralidhar MG, Sanati G, Tankovich T, Kharazi A. High targeted migration of human mesenchymal stem cells grown in hypoxia is associated with enhanced activation of RhoA. Stem Cell Res Ther. 2013;4:5. doi: 10.1186/scrt153
- Dominici M, Le Blanc K, Mueller I, Slaper-Cortenbach I, Marini F, Krause D, et al. Minimal criteria for de?ning multipotent mesenchymal stromal cells. The International Society for Cellular Therapy position state-ment. Cytotherapy. 2006;8:315–317. doi: 10.1080/14653240600855905
- Devine SM. Mesenchymal stem cells: will they have a role in the clinic? J Cell Biochem Suppl. 2002;38:73–79.
- Hil?ker A, Kasper C, Hass R, Haverich A. Mesenchymal stem cells and progenitor cells in connective tissue engineering and regenerative medicine: is there a future for transplantation? Langenbecks Arch Surg. 2011;396:489–497. doi: 10.1007/s00423-011-0762-2
- Williams AR, Hare JM. Mesenchymal stem cells: biology, pathophys-iology, translational ?ndings, and therapeutic implications for cardiac disease. Circ Res. 2011;109:923–940. doi: 10.1161/CIRCRESAHA. 111.243147
- Figueroa FE, Carrión F, Villanueva S, Khoury M. Mesenchymal stem cell treatment for autoimmune diseases: a critical review. Biol Res. 2012;45:269–277. doi: 10.4067/S0716-97602012000300008
- Tsang KS, Ng CPS, Zhu XL, Wong GKC, Lu G, Ahuja AT, et al. Phase I/II randomized controlled trial of autologous bone marrow-derived mesenchymal stem cell therapy for chronic stroke. World J Stem Cells. 2017;9:133–143. doi: 10.4252/wjsc.v9.i8.133
- Savitz SI,Yavagal D, Rappard G, Likosky W, Rutledge N, Graffagnino C, et al.A phase 2 randomized, sham-controlled trial of internal carotid artery infusion of autologous bone marrow-derived ALD-401 cells in patients with recent stable ischemic stroke (RECOVER-Stroke). Circulation. 2019;139:192–205. doi: 10.1161/CIRCULATIONAHA.117.030659
- Cramer SC, Koroshetz WJ, Finklestein SP. The case for modality-speci?c outcome measures in clinical trials of stroke recovery-pro-moting agents. Stroke. 2007;38:1393–1395. doi: 10.1161/01.STR. 0000260087.67462.80
- van de Port IG, Kwakkel G, van Wijk I, Lindeman E. Susceptibility to deterioration of mobility long-term after stroke: a prospective cohort study. Stroke. 2006;37:167–171.
- Dhamoon MS, Moon YP, Paik MC, Boden-Albala B, Rundek T, Sacco RL, et al. Long-term functional recovery after ?rst ischemic stroke: the Northern Manhattan Study. Stroke. 2009;40:2805–2811. doi: 10.1161/STROKEAHA.109.549576
- Wondergem R, Pisters MF, Wouters EJ, Olthof N, de Bie RA, Visser-Meily JM, et al. The course of activities in daily living: who is at risk for decline after ?rst ever stroke? Cerebrovasc Dis. 2017;43:1–8. doi: 10.1159/000451034
- Sennfalt S, Norrving B, Petersson J, Ullberg T. Long-term sur-vival and function after stroke. Stroke. 2019;50:53–61. doi: 10.1161/STROKEAHA.118.022913
- Hsieh YW, Wang CH, Wu SC, Chen PC, Sheu CF, Hsieh CL. Establishing the minimal clinically important difference of the Barthel Index in stroke patients. Neurorehabil Neural Repair. 2007;21:233– 238. doi: 10.1177/1545968306294729
- Detante O, Moisan A, Dimastromatteo J, Richard MJ, Riou L, Grillon E, et al. Intravenous administration of 99mTc-HMPAO-labeled human mes-enchymal stem cells after stroke: in vivo imaging and biodistribution. Cell Transplant. 2009;18:1369–1379. doi: 10.3727/096368909X474230
- Sykova E, Jendelova P. In vivo tracking of stem cells in brain and spinal cord injury. Prog Brain Res. 2007;161:367–383. doi: 10.1016/S0079-6123(06)61026-1
- Shen LH, Li Y, Chen J, Cui Y, Zhang C, Kapke A, et al. One-year follow-up after bone marrow stromal cell treatment in mid-dle-aged female rats with stroke. Stroke. 2007;38:2150–2156. doi: 10.1161/STROKEAHA.106.481218
- Shen LH, Li Y, Chen J, Zacharek A, Gao Q, Kapke A, et al. Therapeutic bene?t of bone marrow stromal cells administered 1 month after stroke. J Cereb Blood Flow Metab. 2007;27:6–13. doi: 10.1038/sj.jcbfm.9600311
- Yasuhara T, Matsukawa N, Hara K, Maki M, Ali MM, Yu SJ, et al. Notch-induced rat and human bone marrow stromal cell grafts reduce ischemic cell loss and ameliorate behavioral de?cits in chronic stroke an-imals. Stem Cells Dev. 2009;18:1501–1514. doi: 10.1089/scd.2009.0011
- Bosetti F, Koenig JI, Ayata C, Back SA, Becker K, Broderick JP, et al. Translational stroke research: vision and opportunities. Stroke. 2017;48:2632–2637. doi: 10.1161/STROKEAHA.117.017112
- Armstead WM, Vavilala MS. Translational approach towards deter-mining the role of cerebral autoregulation in outcome after traumatic brain injury. Exp Neurol. 2019;317:291–297. doi: 10.1016/j.expneurol. 2019.03.015
- Baxter MA, Wynn RF, Jowitt SN, Wraith JE, Fairbairn LJ, Bellantuono I. Study of telomere length reveals rapid aging of human marrow stromal cells following in vitro expansion. Stem Cells. 2004;22:675– 682. doi: 10.1634/stemcells.22-5-675
- Izadpanah R, Kaushal D, Kriedt C, Tsien F, Patel B, Dufour J, et al. Long-term in vitro expansion alters the biology of adult mesenchymal stem cells. Cancer Res. 2008;68:4229–4238. doi: 10.1158/0008-5472. CAN-07-5272
- Galipeau J. The mesenchymal stromal cells dilemma–does a negative phase III trial of random donor mesenchymal stromal cells in steroid-resistant graft-versus-host disease represent a death knell or a bump in the road? Cytotherapy. 2013;15:2–8. doi: 10.1016/j.jcyt.2012.10.002
- Wechsler LR, Bates D, Stroemer P, Andrews-Zwilling YS, Aizman I. Cell therapy for chronic stroke. Stroke. 2018;49:1066–1074. doi: 10.1161/STROKEAHA.117.018290
- Chen J, Chopp M. Exosome therapy for stroke. Stroke. 2018;49:1083– 1090. doi: 10.1161/STROKEAHA.117.018292
- Cramer SC, Sur M, Dobkin BH, O’Brien C, Sanger TD, Trojanowski JQ, et al. Harnessing neuroplasticity for clinical applications. Brain. 2011;134(pt 6):1591–1609. doi: 10.1093/brain/awr039
- Published in Corporate News / Blog
Biodistribution, migration and homing of systemically applied mesenchymal stem/stromal cells
Biodistribution, migration and homing of systemically applied mesenchymal stem/stromal cells
Abstract
Mesenchymal stem/stromal cells (MSCs) are increasingly used as an intravenously applied cellular therapeutic. They were found to be potent in situations such as tissue repair or severe inflammation. Still, data are lacking with regard to the biodistribution of MSCs, their cellular or molecular target structures, and the mechanisms by which MSCs reach these targets. This review discusses current hypotheses for how MSCs can reach tissue sites. Both preclinical and clinical studies using MSCs applied intravenously or intra-arterially are discussed in the context of our current understanding of how MSCs might work in physiological and pathological situations.
Background
In the 1970s, Friedenstein and colleagues [1] first reported that locally applied culture-expanded populations of bone marrow stroma-derived fibroblastic cells remained at their injection sites under the kidney capsule, where an ectopic hematopoiesis was initiated. Later, Arnold Caplan’s group described mesenchymal stem/stromal cells (MSCs) as multipotent mesenchymal cell populations which can differentiate into several tissue types, and demonstrated roles for MSCs in the regeneration of bone, cartilage or ligaments in animal and clinical studies [2–4]. In these studies, however, transplanted cells were followed, if at all, at the site of transplantation, and biodistribution was not an issue.
By the year 2000, clinicians had become increasingly interested in intravenously applied MSCs. Pivotal studies by the group of Horwitz in children with osteogenesis imperfecta, an inherited enzyme deficiency of collagen synthesis by mesenchymal cells in bone, opened the field for intravenous use of MSCs. This concept started from the observation that bone marrow transplantation can provide stromal cells able to synthesize intact collagen type I, replacing deficient patient cell function and ameliorating disease symptoms [5]. Therefore, the authors concluded that transplantation of isolated healthy allogeneic MSCs might cure the disease. This implies homing of transplanted MSCs to sites in bone marrow and/or bone. Efficacy was noted in all six infants treated [5]. Children who received transplants showed improved growth rates and started to synthesize intact bone. Engraftment of donor-type MSC-derived osteoblasts was shown using bone specimens and microsatellite DNA marker analysis. In a second study [6], these authors showed that autologous, enzyme-deficient MSCs transduced with a copy of the intact gene resulted in normal collagen production in bone cavities. Moreover, children who received transplants approached growth curves similar to the children transplanted with allogeneic complete bone marrow [6]. This pioneering work provided the basis for the successful application of MSCs using the intravenous route in other clinical entities.
Establishment of methods to track intravenously administered MSCs
After 2000, the therapeutic use of MSCs by intravenous administration was explored by a number of studies in animals and also humans. These studies used various ways to label culture-expanded MSCs, and to track them in different tissues over time. The tissue source of the MSCs was in most cases not decisive, and cells from various tissue sources were explored. The labeling methodologies used included radioactive labeling of MSCs, labeling with fluorescent vital dyes, contrast agents, transduction with reporter genes, or the use of donor cell-specific DNA markers such as microsatellites [7–11] (reviewed in [12]). The labeling methodologies were, in part, designed to detect only short-term homing of MSCs. In addition, they do not enable the determination of whether detected cells are still alive. These studies were mainly conducted in rodents and nonhuman primates and mostly in non-injury situations. The main common results of these studies were that: MSCs distribute to a variety of tissues after intravenous (i.v.) injection; MSCs are detectable at low or very low frequencies in tissues after transplantation; and signals from the injected cells were found early after administration of the MSCs at the highest frequencies in the lungs, followed by liver and spleen.
The observed biodistribution patterns were confirmed by studies in humans. In patients with mammary carcinoma, Koç et al. [13] demonstrated that i.v. MSCs were well-tolerated in patients at a dose of one million MSCs/kg body weight; however, the cells were trackable in blood only. The data were confirmed in patients with liver cirrhosis using 111In-oxine labeled MSCs, which were found to first accumulate in the lungs followed by continuous increases in liver and spleen up to day 10 after administration [14]. The proportion of accumulation in lung decreased from about 35 % early after transplantation to 2 % or less by day 10, whereas spleen had the highest signals by day 10 after transplant. These results confirm a similar overt biodistribution of MSCs in lung, liver and spleen in humans to that observed in animal models.
Expression of cell adhesion molecules by MSCs as a basis for their interaction with endothelial cells and tissue-directed extravasation
In theory, the main prerequisite for the interaction of transplanted MSCs with endothelial cells are adhesion molecules present on the cell surface of MSCs, and expression of appropriate adhesion counter-receptors on endothelial cells. MSCs (most investigations were performed in human MSCs (hMSCs)) have shown deficits in receptor binding to selectins and/or their ligands. They lack expression of L-selectin, and their E-selectin ligand (CD44) is not functional [15]. MSCs can bind to P-selectin through a fucosylated ligand, which nevertheless is not P-selectin glycoprotein ligand (PSGL)-1 [16]. Thankamony and Sackstein [17] have, however, defined an enzymatic fucosylation procedure which causes the CD44 epitope on MSCs to strongly bind to endothelial E-selectin, resulting in effective rolling of MSCs on endothelial cells and, moreover, extravasation into bone marrow sites. Of the integrins, alpha4beta1 (VLA-4) and alpha5beta1 (VLA-5) have been found to be expressed by MSCs, whereas the beta2 integrins alphaLbeta2 (LFA-1) and alphaMbeta2 (Mac1) could not be detected [15, 16, 18–20] (reviewed in [12, 21]). Interestingly, several chemokine receptors have been found to be expressed on MSCs, including CXCR4, which has been described as a major mediator of the homing and mobilization of hematopoietic cell types [12, 19, 20]. In summary, these findings indicate that MSCs have a deficit with regard to the expressing and/or employing adhesion receptors for coordinated extravasation and tissue-specific homing, as do leukocyte populations.
Emergence of common themes in exploring the biodistribution of MSCs
Subsequent to the first reports on the homing and migration of transplanted MSCs to tissues, additional questions about MSC biodistribution have been addressed, including quantification of MSCs, their preferential homing to several target sites, and the involvement of cues, such as regeneration or inflammation, and the size of MSCs in determining their biodistribution (Table 1).
Table 1
Common themes in MSC biodistribution research
| Theme | Targeted tissues (possible mechanism) | References |
| Increased homing after intra-arterial delivery compared with intravenous delivery? | Kidney | [33, 34] |
| Joints | [32] | |
| Stroke | [30] | |
| Other (many) tissues | [31] | |
| Side effects of intra-arterial versus intravenous delivery? | Incorporation into vessel wall | [23, 35] |
| Obstruction of microvessels | [38] | |
| Vascular occlusion | [39] | |
| Targeting of vessel wall and vessel-associated tissues? | Lungs, lymph nodes, intestine | [47] |
| Targeting of tissues for regeneration | Myocardium | [18, 48–55] |
| Beta1 integrins | [48, 49] | |
| CCL2, monocytes | [52] | |
| Kidney | [33, 56–63] | |
| Gut and liver | [64–67] | |
| Skin | [44, 68–71] | |
| CCL21 | [44] | |
| JAM-A | [68] | |
| Brain | [72–75] | |
| P/E selectin (CD44) | [73] | |
| CXCR4/flk-1/EPO-R | [74] | |
| Homing to bone marrow | Bone marrow | [76–81] |
| HCELL/E-selectin | [15] | |
| Subendothelial localization | [79] | |
| Biodistribution to the immune system? | Macrophages | [37, 41, 42] |
| Dendritic cells | [38] | |
| T cells | [39] | |
| Unknown target cells | ||
| Idoleamine desoxygenase | [43] | |
| Prostaglandin E2 | [37, 41] | |
| Elimination mechanisms? | Antibody formation | [6] |
| Phagocytes | [102] | |
| Influence of radiation on homing? | Increased in brain, heart, bone marrow, and muscles | [43, 82] |
| Homing in malignancies? | Tumor | [83–85, 87–92] |
| Mediated by CCL25 | [88] | |
| Mediated by sodium iodide symporter under the control of RANTES/CCL-5 promoter | [87] | |
| Homed MSCs form tumor-associated fibroblasts | [90] | |
| Formation of microvesicles | Microvesicles may contribute to/be part of MSC biodistribution | [14, 63, 93–97] |
| Mediated by horizontal transfer of microRNAs | [96] |
In many of the earlier studies, the target sites as well as the molecular mechanisms governing the interactions of MSCs with the local environment after transplantation (e.g., endothelial cells, target tissue), such as adhesion molecules or signaling mechanisms, were either not addressed or were analyzed only to a minor degree. Moreover, MSCs were often evaluated by microscopy, a method relatively prone to artifacts. Many studies also did not quantify the numbers of MSCs in target or other tissues. Likewise, only few studies reported on the size of the identified MSCs. Despite this lack of information, other themes have emerged, especially research on cues that may regulate the biodistribution of systemically applied MSCs; these include first pass tissues, specifically the lungs, inflammation, irradiation, sites of hypoxia or repair, and cancer (Table 1). As a result, concepts have been raised which imply an ability of MSCs to migrate to specific sites—e.g., MSCs as an “injury drugstore” for several acute clinical situations [21, 22].
First-line accumulation of intravenously administered MSCs in the lungs
The first hurdle for intravenously transplanted MSCs is the lung capillary bed. After culture expansion, MSCs are relatively large cells with an estimated average size of around 30 µm in suspension (ranging from 16–53 µm) [23]. Their size may also vary depending on the osmolarity of the culture media, passage number, and/or cell density during seeding as well as general culture conditions (two-dimensional versus three-dimensional culture). In comparison with MSCs, hematopoietic stem cells have a much smaller diameter, ranging from 4–12 µm depending on the subfraction analyzed [24, 25]. Therefore, obstructive events during lung passage are expected after intravenous administration of MSCs. Lee et al. [26] presented a kinetic study of MSCs accumulating in murine lungs in which up to 80 % of injected cells were found in the lungs within a few minutes after injection. Moreover, formation of emboli in lung vessels was noted. The MSC signal (an Alu sequence DNA marker) fell exponentially, with a half-life of about 24 h and practically complete disappearance after 4 days [26]. Barbash and colleagues [10] confirmed the detection of the overall MSC load in the lungs using 99mTc-labeled MSCs in a rat model with induced myocardial infarction. Murine MSCs also showed deleterious effects in mice, including post-injection lethality, which was not the case after administration of hMSCs [27]. Interaction of human or murine MSCs with lung endothelial cells was dependent on the suspension medium in which the transplanted cells were administered [27]. Adhesion of the MSCs to endothelial cells was found to involve the integrin ligand vascular cell adhesion molecule (VCAM)-1. When comparing MSCs with mononuclear cells from bone marrow, neural stem cells and multipotent adult progenitor cells, Fischer et al. [28] found that MSCs showed the highest interaction with lung endothelia, which could be inhibited by pretreatment with anti-CD49d antibody. In a study by Kerkelä et al. [29], adhesion of MSCs to lung tissue (probably endothelial cells) was dependent on the enzyme treatment used during harvesting of confluent MSCs in culture before transplantation; after treatment with pronase, MSCs more readily cleared the lungs and could be found in other tissues compared with trypsinization treatment. Taken together, these data indicate an active role of the adhesion molecules VLA-4/VCAM-1 on MSCs/endothelial cells during interaction of MSCs with lung tissue. It remains to be clarified, however, whether this is a passive or active process. Also, relatively little is known about possible adhesion molecules other than VLA-4/VCAM-1 which may be operative in the interaction of MSCs with endothelial cell surfaces in the lung. This includes the fucosylation of CD44 to HCELL, a highly active E-selectin ligand on MSCs, which is relevant in bone marrow endothelia but seemingly did not affect lung interactions [15].
In summary, presently there is strong evidence that accumulation of MSCs in the lungs is a key determining factor for their biodistribution. The major adhesion molecule involved seems to be VLA-4/VCAM1. Still, it is not clear to what degree the findings in animal studies are quantitatively transferable to humans (Table 1).
Biodistribution of MSCs after intra-arterial versus intravenous administration
Studies comparing intra-arterial and intravenous application of MSCs have demonstrated a major association between intravenous application and retention of MSCs in the lungs, and their increased accumulation in therapeutic target tissues after intra-arterial injection. Walczak et al. [30] in a rat transient ischemia stroke model applied two independent detection methods (magnetic resonance imaging and Doppler flowmetry). They demonstrated that higher cerebral engraftment rates are associated with impeded cerebral blood flow, and that intra-arterial delivery may be advantageous in ischemic stroke to deliver MSCs to the site of injury. Mäkelä et al. [31] compared intra-arterial and intravenous administration of MSCs labeled with 99mTc, and also found that the intra-arterial transplantation route has a positive impact on the biodistribution of bone marrow-derived MSCs (BM-MSCs) to peripheral tissues. They found that intra-arterial transplantation decreased the deposition of BM-MSCs in the lungs and increased uptake in other organs, especially in the liver. In a study looking at human adipose tissue-derived MSCs in SCID mice, Toupet et al. [32] showed that 15 % of intra-arterially injected MSCs accumulate in inflamed joints during the first month, and 1.5 % over a longer term of >6 months, also favoring intra-arterial over intravenous application for, in their case, anti-inflammatory MSCs. Therapeutic effects of MSCs in kidney have been generally achieved after intra-arterial delivery [33, 34]. Although more studies will be needed, these data suggest that the intra-arterial route of administration is effective in avoiding pulmonary entrapment of BM-MSCs, and may thus improve the biodistribution and bioavailability of transplanted MSCs in clinically relevant tissues for, e.g., tissue repair.
Interactions of MSCs with the blood vessel wall: integration into the vessel wall or transmigration?
As described above, the majority of intravenously injected MSCs are generally detected in the lungs, and in no other tissue at comparable numbers even at later time points. Some groups asked whether MSCs may directly target vessels or perivascular tissue and investigated the fate of MSCs in and around blood vessels. These studies followed the cells using intravital microscopy and histologic examination in different tissues after intra-arterial [23, 30, 35] administration. In the cremaster muscle intravital microscopy model, Furlani et al. [23] observed that the microcirculation was disturbed, with some MSCs obstructing small vessels. In addition, pulmonary emboli were found. Toma et al. [35] also observed occlusion of microvessels and entrapment of the injected MSCs. Moreover, they observed stable integration of some transplanted cells into the vessel wall. Cui et al. [36] reported a risk of vascular occlusion in their rat stroke infarction model after intra-arterial injection, pointing to the fact that local intravasal entrapment of MSCs may frequently occur, and MSCs may obstruct the microcirculation. Currently, however, we lack conclusive data that MSCs that are entrapped in capillaries and/or are incorporated into the vessel wall or adjacent to endothelial cells would relocate (i.e., “home”) to their main tissue of origin, pericytes.
Transplanted MSCs interact with cells of the immune system
Transplanted MSCs have been shown to rapidly interact with immune cell types, which are—at least in part—present also in the bloodstream. In a lung sepsis model, Nemeth et al. [37] observed that MSCs co-localize with lung-resident macrophage cells and induce them to produce anti-inflammatory interleukin (IL)-10 via release of prostaglandin E by MSCs as part of their therapeutic effect. Chiesa et al. [38] showed that interstitial dendritic cells (DCs) decrease their physiological migration from skin to lymph nodes rapidly after intravenous administration of MSCs. They describe that MSCs inhibit Toll-like receptor (TLR)-4-induced activation of DCs, which results in the inhibition of cytokine secretion by DCs, downregulation of adhesion molecules involved in the migration of DCs to the lymph nodes, suppression of DC antigen presentation to CD4+ T cells, and cross-presentation to CD8+ T cells. Akiyama et al. [39] demonstrated that both human and murine MSCs can induce immune suppression by attracting and killing autoreactive T cells through FasL, thereby stimulating transforming growth factor beta production by macrophages and generation of regulatory T cells. The interaction has been shown to involve the secretion of MCP-1 by MSCs. The dying T cells in turn activate macrophages to produce transforming growth factor beta, thus stimulating regulatory T cells and promoting immune tolerance. Possibly, the secretion of anti-inflammatory protein TSG-6 by activated MSCs, which has been described in a zymosan-induced mouse peritonitis model, involves an interaction via TLR2/reduction of NF-?B signaling in resident macrophages [40].
Another type of potential interaction between MSCs and immune cells is suggested by data from Kim et al. [41], who used an in vitro system showing that murine MSCs inhibit functionality of DCs through TLR-4-mediated signals in co-culture with monocytes. During this study, hMSCs revealed a unique immunophenotype of alternatively activated human monocytes which are CD206-high, IL-10-high, IL-6-high, IL12-low, and tumor necrosis factor (TNF)-alpha-low [42]. The immune suppressive effects of MSCs have been shown to depend on induction of indoleamine 2,3-dioxygenase [43], or production of prostaglandin E2 as a main effector to dampen inflammation [37, 44]. These data indirectly support the hypothesis that MSCs interact directly with monocytic and/or antigen-presenting cells in vivo.
The successful therapeutic use of MSCs in patients with severe immune dysregulations, such as graft-versus-host disease after allogeneic hematopoietic stem cell transplantation, has attracted high interest by hematologists (reviewed in [45]). The studies were based on a number of in vitro findings that MSCs can either interact with or affect the function of various types of immune effector cells such as antigen-presenting cells, B or T lymphocytes, or natural killer (NK) cells (reviewed in [46]). In all these studies, identification of MSCs at target sites has been cumbersome, and often no transplanted MSCs were identified. von Bahr et al. [47] reported that MSC donor DNA was detectable at low levels in 8 out of 18 patients in vessel-associated tissues in the patients, including lungs, lymph nodes, and intestine. Detection of MSC donor DNA was negatively correlated with time from infusion to sample collection.
Together, these studies strongly indicate the existence of interactions between transplanted MSCs and cells of the immune system. This way, MSCs also biodistribute to the immune system through contact with different types of leukocytes in the circulation or various tissues such as skin, spleen, and lymph nodes.
Potential mechanisms of elimination of MSCs from the circulation
A relevant aspect of the interaction between transplanted MSCs and immune system cells, in both animal models and humans, is the induction of xenogeneic and allogeneic immune responses, resulting in antibody formation or T-cell responses against the transplanted MSCs. Induction of antibody formation explains the failure to identify transplanted MSCs in patients upon repeated administration of allogeneic MSCs that had been cultured in fetal bovine serum-containing media [6]. Anti-fetal calf serum antibody formation has been demonstrated in patients that did not respond to repeated MSC applications [6]. Elimination of xenogeneic MSCs in some of the animal models studied may occur in ways analogous to those in the allogeneic situation.
Despite the fact that several target tissues of MSCs have been established, there are few data as to the place to which systemically applied MSCs will finally migrate, or where they end up before or when they are eliminated. The fact that the transplanted MSCs are often not detectable at all, or only a small fraction of them is traced, underscores the potential relevance of the lung as a “first pass” tissue, and may indicate an involvement of lung trapping in elimination of MSCs. On the other hand, the fact that MSCs are barely or not at all detectable in patients after transplantation demonstrates that systemic pathways to eliminate transplanted MSCs may be operating in humans, leading to barely detectable long-term engraftment.
Tissue repair situations which provide cues to attract transplanted MSCs
The interactions of MSCs with different types of immune cells point to their ability to respond to signals from the immune system. Since aspects of tissue repair have been associated with (adaptive) immune responses, it is likely that inflammatory and tissue repair signals influence MSC responses in vivo, including their biodistribution.
Myocardial infarction
The VLA-4/VCAM receptor axis has been shown to be involved in MSC migration in myocardial infarction. Pre-treatment of MSCs with TNF-1alpha stimulated migration of MSCs through heart endothelia mediated through VCAM-1, indicating that beta1 integrins are actively involved in this process [48]. Confirming this hypothesis, Ip et al. [49] demonstrated in a murine model that alpha4 integrin is required for migration of MSCs to myocardium, whereas the chemokine receptor CXCR4 was dispensable for the entry of transplanted cells into ischemic tissue.
Intravenously administered MSCs have been observed to, at least transiently, accumulate in areas of myocardial ischemia [18, 50, 51]. To this end, Belema-Bedada et al. [52] employed a transgenic mouse model expressing the monokine CC-chemokine ligand (CCL)2 under a cardiac specific promotor, increasing CCL2 expression in heart muscle. These authors observed that i.v. MSCs accumulate rapidly and selectively in the heart. They showed that the migration of the MSCs to heart is preceded by monocyte emigration to the myocardium, and involves G-protein-coupled receptors, pointing also towards the involvement of chemokine signals. Kraitchman et al. [11] confirmed the accumulation of i.v. MSCs into myocardial infarction areas using a radioimaging tracer and single-photon emission computed tomography in a dog model. Wang et al. [53] traced MSCs at later stages after infarction, and saw markers of newly regenerated cardiomyocytes. It is also not clear whether MSCs steadily incorporate into cardiac tissue. Other studies have failed to detect any homed MSCs in cardiac tissue over the long term (e.g., [54]). Jasmin et al. [55] injected MSCs i.v. after nanoparticle labeling in a model of heart inflammation caused by the Chagas disease parasite Trypanosoma cruzi. They observed that although most MSCs migrated to the lungs, liver and spleen, a few cells homed to the inflamed heart. In conclusion, some mechanisms seem to recruit, mostly transiently, some MSCs to inflamed or ischemic heart, including VLA-4/VCAM-1 and the CCL2 and possibly other chemokine receptor signals.
Kidney damage
Despite the wide range of beneficial effects seen with the therapeutic use of MSCs in animal models, only a few clinical trials have tested the efficacy of MSCs for renal diseases. Reinders and colleagues [56] used intravenous injection of 1?×?106 autologous BM-MSCs/kg in six kidney allograft recipients to dampen rejection of the graft and/or decrease interstitial fibrosis and tubular atrophy. Likewise, Tan et al. investigated autologous BM-MSCs (1–2?×?106/kg) at kidney reperfusion and 2 weeks after application; the incidence of acute rejection decreased and renal function at 1 year improved compared with anti-IL-2 receptor antibody induction therapy [57]. In a clinical phase I safety trial, five patients aged >65 years with underlying renal disease and multiple comorbidities were infused with allogenic MSCs during coronary artery bypass or cardiac surgery. Although the follow-up period was short and one of the patients died, none of the patients required dialysis, supporting the beneficial influence of MSCs on repair of kidney damage [58, 59].
In animal studies, MSCs were also associated with repair of the permeability barrier of the glomerulus in an Alport disease model [60] and improved kidney function in an experimental sepsis mouse model through reprogramming of macrophages via release of prostaglandin E2 [37]. Morigi and colleagues [61, 62] have shown that treatment with murine BM-MSCs (2?×?105 per mouse) in an acute renal failure mouse model induced by cisplatin (a nephrotoxic anti-cancer drug) protected the animals from renal function impairment and tubular injury. Intriguingly, the effects of MSCs in stimulating proliferation and inhibiting apoptosis of tubular epithelial cells in a glycerol-induced acute kidney injury SCID mouse model could also be achieved by using microvesicles derived from hMSCs [63]. In addition to these human studies, several studies demonstrate that MSCs localize within injured kidneys when injected in mice with acute kidney injury (e.g., [34, 63]; reviewed in [58]). The presence of MSCs at later stages of kidney injury or regeneration has not been studied, but the therapeutic benefits have been measured, and intra-arterial injection of MSCs seems to be more favorable [33, 34, 61].
Liver damage
Gholamrezanezhad et al. [14] studied i.v. infused 111In-oxine-labeled MSCs in patients with liver cirrhosis. The radioactivity was first observed to accumulate in the lungs. During the following hours to days, the radioactivity gradually increased in the liver and spleen, with spleen uptake exceeding that in the liver in all patients. In the liver and spleen, radioactivity increased by day 10 post-infusion, whereas residual activity in the lungs decreased approximately tenfold. In contrast, Briquet et al. [64] saw no recruitment of hMSCs to liver damaged by CCl4 intoxication in immune-deficient mice. A study by Zhang et al. [65] indicates that corticosteroids and the SDF-1/CXCR4 axis are involved in MSC migration in a carbon tetrachloride-induced liver fibrosis model. Another liver regeneration model in mice indicated that MSC homing to liver was associated with regeneration, but the mechanisms for this were not investigated [66]. In summary, although many of the published studies have not addressed aspects of MSC biodistribution, there is some evidence for biodistribution to injured or diseased livers, but the underlying mechanisms are mostly unclear.
Gut and skin
Only a few studies have analyzed MSC accumulation in epithelial tissues so far. Inflammatory bowel disease models have addressed homing of i.v. MSCs. Parekaddan et al. [67] demonstrated the presence of MSC-derived signals not only in lungs and spleen but also in the gut of affected animals. Sasaki et al. [44] assessed whether homed MSCs can differentiate into skin cells, including keratinocytes, and whether they could contribute to wound repair. They i.v. injected green fluorescence protein (GFP) transgenic MSCs and identified GFP-positive cells associated with specific markers for keratinocytes, endothelial cells, and pericytes. They attribute the extravasation to inflamed areas to the presence of the chemokine CCL21 in vessels in the inflamed tissue. Still, numbers of detected MSCs in the wounded skin areas were low. MSCs have been found in wound tissues several days after transplantation in animal models [68–71] but their engraftment efficiency ranged from <0.01 % when MSCs were intravenously injected to 3.5 % in a study where MSCs were locally applied. This points to a minor role of i.v. injected MSCs in skin repair. One study reported that, after intravenous injection of GFP transgenic MSCs, keratinocytes, endothelial cells, pericytes and macrophages within the healed wound were all found to be GFP-positive. The authors concluded that they might be derived from donor MSCs [71].
Brain
Some studies have investigated whether transplanted MSCs migrate into inflamed brain tissue. In murine stroke models, MSCs migrated into ischemic areas after intravenous delivery [72, 73]. The latter study mentions that the MSCs are recruited to these sites via endothelial expressed P- and E-selectin, and that CD44 is present on the MSCs. In their rat brain ischemia model, Wei et al. [74] found that i.v. MSCs localize to ischemic zones and deliver neurotrophic factors. This occurs at an increased rate when MSCs have been exposed to hypoxia before transplantation. The extravasation efficiency of the MSCs correlated with increased expression of CXCR4, flk-1 and the erythropoietin receptors, and downregulation of pro-inflammmatory regulators in the homing MSCs. The activity of microglia formation was suppressed in animals after MSC therapy, and NeuN-positive and Glut1-positive cells were increased. Constantin et al. [75] used intravital microscopy in a murine experimental autoimmune encephalitis model. They found, using bioluminescence, accumulation of a subset of transplanted MSCs in inflamed brain venules in inflammatory foci of experimental autoimmune encephalomyelitis 16 and 30 days after transplantation, and showed a role for alpha4 integrin in the migration process of MSCs into brain tissue. Although absolute numbers of transplanted MSCs were not determined and may be low, the results indicate that active inflammation may switch the homing behavior of transplanted MSCs from unspecific entrapment to specific recruitment.
Together, these data indicate that MSCs can migrate into ischemic and proinflammatory regions in certain disease models. Mostly short- (within the first 3 days) and mid-term (3 days to 3 months) homing has been reported, whereas long-term persistence (>3 months) of MSCs is rarely detected. Due to the technologies used to detect transplanted cells, there is only limited evidence to indicate whether the MSCs home as intact cells into their target environments. The data are in favor of transient homing and locally acting MSCs in the investigated pathologies.
Homing of transplanted MSCs to bone marrow
Several decades of clinical and experimental work in the field of bone marrow transplantation have shown that donor type MSCs will generally not engraft in allogeneic hosts, including the precursor cell type for MSCs, fibroblast colony-forming units [76–78]. Rombouts and Ploemacher [79] demonstrated that prolonged time in culture induces a defect in MSCs that affects their engraftment into bone marrow in a classic bone marrow transplantation situation. In contrast, as reported above, Horwitz and colleagues [5, 6] demonstrated that MSCs engraft into bone marrow of children with osteogenesis imperfecta. Possibly, engraftment of MSCs therefore requires a “niche” which is not free in normal bone marrow transplant recipients, but is created in a deficiency state such as the collagen synthase defect found in osteogenesis imperfecta. Follenzi et al. [80] recently demonstrated that mice suffering from hemophilia A, when transplanted with normal healthy total bone marrow cells, show engraftment not only of hematopoietic cells but also of subendothelial MSC-like cells. Interestingly, these MSCs had not been cultured before transplantation. Functional MSCs may, therefore, engraft, at least in the case of certain deficiencies in the transplanted hosts. Interestingly, the group of Horwitz more recently showed that non-plastic-adherent bone marrow cells engraft in a murine model and give rise to osteoprogenitors, which are more potent osteoprogenitors than “classic” plastic-adherent MSCs in mice [81]. This underscores the possibility that the culture period induces the engraftment defect, and that, in addition, cells other than “classic” MSCs can mediate stromal engraftment. On the other hand, “classic” plastic-adherent MSCs have been shown to remain as a source of hematopoietic environment when transplanted into tissues other than bone marrow [1]. In contrast to these findings, the model by Sackstein et al. [15], where an active E-selectin ligand was engineered on the surface of plastic-adherent MSCs, resulted in efficient homing to bone marrow, indicating the possibility of BM-MSCs (or MSCs from other tissue sources) distributing to bone marrow.
Influence of irradiation on migration and biodistribution of MSCs
In a murine study, Francois et al. [43] showed that both total body irradiation and local irradiation (e.g., selective irradiation of abdomen or legs) affected the distribution of i.v. infused hMSCs in NOD/SCID mice compared with untreated animals. Intravenously infused hMSCs were found only in minimal amounts exclusively in the lung, bone marrow, and muscles in non-irradiated control animals. Mice after total body irradiation had increased absolute numbers of hMSCs in brain, heart, bone marrow, and muscles. Moreover, selective radiation of limbs or the abdomen yielded increased engraftment of hMSCs in the exposed skin or muscles than with total body irradiation alone. hMSC engraftment outside the locally irradiated regions was also increased, pointing to both local and systemic effects of irradiation on MSC engraftment. The study did not investigate long-term engraftment. Sémont et al. [82] looked at the engraftment and efficacy of transplanted MSCs in an immunodeficient mouse model of radiation-induced gastrointestinal tract failure. They demonstrated accelerated recovery in the group receiving hMSCs, with decreased apoptosis of epithelial cells and increased proliferation within the small intestinal mucosa. Yet, transplanted MSCs were not detected at significant amounts.
A special case: migration and engraftment of MSCs into tumors
Tumor-associated fibroblasts have been described as a form of MSCs, which are recruited from the MSC pool and are an integral part of the microenvironment of many different solid tumors [83, 84]. Tumor tissue therefore also represents a target for the homing of i.v. injected MSCs. In experimental studies, both beneficial and adverse effects have been reported. Beckermann et al. [85] verified the migration of i.v. MSCs into areas close to the vessel wall in human pancreatic tumors in immunodeficient mice. Alieva et al. [86] followed locally implanted adipose tissue-derived MSCs with a genetic modification induced by lentiviral transduction and traced them by bioluminescence in a glioblastoma model. After incorporation of the transplanted MSCs, administration of gancyclovir activates the thymidine kinase transgene, resulting in death and elimination of the transplanted MSCs and tumor regression. A PECAM-Promotor-driven second transgene as reporter construct served to indicate that the transplanted MSCs can acquire endothelial-like characteristics. Similarly, Knoop et al. [87] used i.v. MSCs expressing sodium iodide symporter under the control of the RANTES/CCL-5 promoter; when loaded with 131I compound these conferred significant anti-tumor effects.
Xu et al. [88], in a myeloma model, showed that MSCs are chemoattracted by the chemokine CCL25, thus supporting myeloma growth. In a Ewing sarcoma nude mouse model, i.v. injected MSCs expressing IL-12 were effective in treating the sarcomas [89]. Interestingly, the transplanted MSCs themselves were not identified, while the secreted IL-12 was. Kidd et al. [90] showed that tumor-associated fibroblasts originating from transplanted MSCs in syngeneic ovarian and breast cancers are recruited from the bone marrow, whereas the bulk of the vascular and fibrovascular stromal cells (pericytes, a-smooth muscle actin-positive myofibroblasts, and endothelial cells) were recruited from adipose tissue. These data indicate a process whereby, once bone marrow homing of transplanted MSCs is established, these MSCs may be (genetically) directed along pre-established pathways of endogenous MSCs that circulate from bone marrow to the tumor. Further work by Grisendi et al. [91] demonstrated that the process of MSC incorporation into tumors implies the formation of epithelial–mesenchymal or endothelial–mesenchymal transitions, and requires the formation of fibroblasts derived from mesenchymal progenitors.
MSCs were also found to enhance angiogenesis, as shown in models of B16 melanoma cells and Lewis lung carcinoma [92]. Co-injection of tumor cells and MSCs led to increased tumor size compared with injection of tumor cells alone. Tumor vessel areas were greater in tumors after co-injection of tumor cells with MSCs than in tumors induced by injection of cancer cells alone. Co-injected MSCs localized close to vascular walls, and also expressed the endothelial marker CD31/PECAM-1.
In conclusion, MSCs show a clear tumor tropism. Many data indicate that they are incorporated into the tumor microenvironment and can stimulate tumor growth. Their biodistribution and tumor tropism, however, may also be exploited to target tumors, e.g., using a suicide transgene approach.
Recent developments: exosomes, microparticles and MSCs
As with many other cell types, MSCs are capable of forming exosomes [63, 93, 94]. Exosomes are small membrane vesicles (40–100 nm in diameter) of endosomal origin derived from MSCs. Exosomes have been found to accumulate in target cells of MSC therapy, such as tubular cells in acute kidney injury [63], or after recovery from traumatic brain injury [95]. In other studies, microvesicles have been found to contain signaling molecules which are hypothesized to be important for MSC-mediated therapeutic effects by horizontal transfer, such as miR-133b in a rodent stroke model [96], or insulin-like growth factor receptor in renal tubular injury [97]. Kordelas et al. [98] administered exosomes isolated from MSCs to a patient with severe graft-versus-host disease; this patient showed marked improvement after the exosome infusion. This field is currently expanding rapidly, and can only be covered briefly by this review. One of the relevant open questions for the biodistribution of MSCs is whether exosomes are indeed formed by intravasally administered MSCs.
Summary: possible ways for MSCs to interact within the local environment of the bloodstream to direct their biodistribution
A summary of the possible ways MSC might interact within the blood circulation is shown in Fig. 1. MSC surface marker profiling has revealed no expression of the co-stimulatory molecules CD40, CD86, and CD80 needed for correct T-cell responses leading to T-cell anergy. In vitro studies also showed that CD4+ T cells in contact with MSCs were arrested in the G1/G0 phase and stopped proliferating whereas regulatory T cell proliferation was favored and IgG production by plasma cells seemed to be affected [46]. In addition, MSCs only express a low amount of major histocompatibility complex (MHC) I and almost no MHC II (except after interferon-? treatment), making them more evasive to NK cell cytoxicity in an allogenic/xenogenic setting. Interactions between NK cells and MSCs in general have been controversial, as discussed by different groups (e.g., [99–101]). MSCs seem to lower NK cell cytoxicity through downregulation of interferon-? expression and production of anti-inflammatory IL-4 and IL 10, but NK cells were associated with the ability to lyse MSCs from allogenic donors [99]. Additionally, the so-called instant blood-mediated inflammatory response might be triggered by the innate immune response caused by tropism of dying MSCs within the blood circulation, resulting in complement activation and opsonization of injected MSCs following uptake of marked MSC cell fragments by primary/secondary phagocytes, as was shown by Moll et al. [102]. Intravital microscopy of MSCs in a cremaster muscle mouse model (our unpublished data) revealed that MSCs are likely to be disrupted by the shear force of the blood flow, resulting in fragmentation of the cell and creation of small extracellular vesicles able to influence paracrine secretion of immunomodulatory molecules or cause phagocytosis of these fragments by macrophages and endothelial cells, subsequently followed by clearance of disrupted MSCs in the liver and spleen within a few days. MSCs that find a niche and survived the journey through the bloodstream might interact actively or passively with the endothelial wall and may extravasate after interacting with the extracellular matrix (e.g., with MMP 2 and gelatinase) and reside in a pericyte-like location in the long term.
Conclusion
The final fate of the bulk of i.v. injected MSCs remains elusive, since preclinical animal studies and some human data have been able to detect only small proportions, if any, of injected MSCs. A number of open questions remain. These include: Which contacts are made between MSCs and other cells upon infusion in the bloodstream and what are the consequences of these? What is the fate of MSCs that do not migrate into inflamed tissue and are there physiological clearance pathways for transplanted MSCs? Given that many therapeutic effects have been observed without detectable MSCs in the target tissues, are intact MSCs therefore relevant for the observed effects?
We believe that further careful analysis of animal disease models, including investigation of the role of mediators such as exosomes, signaling proteins, and microRNAs, will help further advance our understanding of why we have so far not obtained clear answers about how MSCs biodistribute, migrate and home, and how these cells exert their beneficial effects, and what might be the potential of these new insights for the development of further improvements of MSC-derived therapies.
Note
This article is part of a thematic series ‘Mesenchymal Stem/Stromal Cells—An update’. Other articles in this series can be found at http://www.biomedcentral.com/series/mesenchymal
Acknowledgments
The authors thank the team of the Institute of Transfusion Medicine and Immune Hematology, German Red Cross Blood Donor Service, University of Frankfurt for their constant support.
Abbreviations
| BM-MSC | Bone marrow-derived mesenchymal stem/stromal cell |
| DC | Dendritic cell |
| GFP | Green fluorescence protein |
| hMSC | Human mesenchymal stem/stromal cell |
| i.v. | Intravenous/intravenously |
| IL | Interleukin |
| MHC | Major histocompatibility complex |
| MSC | Mesenchymal stem/stromal cell |
| NK | Natural killer |
| TLR | Toll-like receptor |
| TNF | Tumor necrosis factor |
| VCAM | Vascular cell adhesion molecule |
Footnotes
Competing interests
The authors declare that they have no competing interests.
Authors’ contributions
All authors read and approved the final manuscript.
References
- Friedenstein AJ, Chailakhyan RK, Latsinik NV, Panasyuk AF, Keiliss-Borok IV. Stromal cells responsible for transferring the micro-environment of the hemopoietic tissues. Cloning in vitro and retransplantation in vivo. Transplantation. 1974;17:331–40. doi: 10.1097/00007890-197404000-00001. [PubMed] [CrossRef] [Google Scholar]
- Pittenger MF, Mackay AM, Beck SC, Jaiswal RK, Douglas R, Mosca JD, et al. Multilineage potential of adult human mesenchymal stem cells. Science. 1999;284:143–7. doi: 10.1126/science.284.5411.143. [PubMed] [CrossRef] [Google Scholar]
- Caplan AI, Dennis JE. Mesenchymal stem cells as trophic mediators. J Cell Biochem. 2006;98:1076–84. doi: 10.1002/jcb.20886. [PubMed] [CrossRef] [Google Scholar]
- Caplan AI. Why are MSCs therapeutic? New data: new insight. J Pathol. 2009;217:318–24. doi: 10.1002/path.2469. [PubMed] [CrossRef] [Google Scholar]
- Horwitz EM, Gordon PL, Koo WK, Marx JC, Neel MD, McNall RY, et al. Isolated allogeneic bone marrow-derived mesenchymal cells engraft and stimulate growth in children with osteogenesis imperfecta: implications for cell therapy of bone. Proc Natl Acad Sci U S A. 2002;99:8932–7. doi: 10.1073/pnas.132252399. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Horwitz EM, Prockop DJ, Fitzpatrick LA, Koo WW, Gordon PL, Neel M, et al. Transplantability and therapeutic effects of bone marrow-derived mesenchymal cells in children with osteogenesis imperfecta. Nat Med. 1999;5:309–13. doi: 10.1038/6529. [PubMed] [CrossRef] [Google Scholar]
- Devine SM, Bartholomew A, Mahmud N, Nelson M, Patil S, Hardy W, et al. Mesenchymal stem cells are capable of homing to the bone marrow of non-human primates following systemic infusion. Exp Hematol. 2001;29:244–55. doi: 10.1016/S0301-472X(00)00635-4. [PubMed] [CrossRef] [Google Scholar]
- Devine SM, Cobbs C, Jennings M, Bartholomew A, Hoffman R. Mesenchymal stem cells distribute to a wide range of tissues following systemic infusion into nonhuman primates. Blood. 2003;101:2999–3001. doi: 10.1182/blood-2002-06-1830. [PubMed] [CrossRef] [Google Scholar]
- Gao J, Dennis JE, Muzic RF, Lundberg M, Caplan AI. The dynamic in vivo distribution of bone marrow-derived mesenchymal stem cells after infusion. Cells Tissues Organs. 2001;169:12–20. doi: 10.1159/000047856. [PubMed] [CrossRef] [Google Scholar]
- Barbash IM, Chouraqui P, Baron J, Feinberg MS, Etzion S, Tessone A, et al. Systemic delivery of bone marrow-derived mesenchymal stem cells to the infarcted myocardium: feasibility, cell migration, and body distribution. Circulation. 2003;108:863–8. doi: 10.1161/01.CIR.0000084828.50310.6A. [PubMed] [CrossRef] [Google Scholar]
- Kraitchman D, Tatsumi M, Gilson WD, Ishimori T, Kedziorek D, Walczak P, et al. Dynamic imaging of allogeneic mesenchymal stem cells trafficking to myocardial infarction. Circulation. 2005;112:1451–61. doi: 10.1161/CIRCULATIONAHA.105.537480. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Karp JM, Leng Teo GS. Mesenchymal stem cell homing: the devil is in the details. Cell Stem Cell. 2009;4:206–16. doi: 10.1016/j.stem.2009.02.001. [PubMed] [CrossRef] [Google Scholar]
- Koç ON, Gerson SL, Cooper BW, Dyhouse SM, Haynesworth SE, Caplan AI, et al. Rapid hematopoietic recovery after coinfusion of autologous-blood stem cells and culture-expanded marrow mesenchymal stem cells in advanced breast cancer patients receiving high-dose chemotherapy. J Clin Oncol. 2000;18:307–16. [PubMed] [Google Scholar]
- Gholamrezanezhad A, Mirpour S, Bagheri M, Mohamadnejad M, Alimoghaddam K, Abdolahzadeh L, et al. In vivo tracking of 111In-oxine labeled mesenchymal stem cells following infusion in patients with advanced cirrhosis. Nucl Med Biol. 2011;38:961–7. doi: 10.1016/j.nucmedbio.2011.03.008. [PubMed] [CrossRef] [Google Scholar]
- Sackstein R, Merzaban JS, Cain DW, Dagia NM, Spencer JA, Lin CP, et al. Ex vivo glycan engineering of CD44 programs human multipotent mesenchymal stromal cell trafficking to bone. Nat Med. 2008;14:181–7. doi: 10.1038/nm1703. [PubMed] [CrossRef] [Google Scholar]
- Ruster B, Gottig S, Ludwig RJ, Bistrian R, Muller S, Seifried E, et al. Mesenchymal stem cells display coordinated rolling and adhesion behavior on endothelial cells. Blood. 2006;108:3938–44. doi: 10.1182/blood-2006-05-025098. [PubMed] [CrossRef] [Google Scholar]
- Thankamony SP, Sackstein R. Enforced hematopoietic cell E- and L-selectin ligand (HCELL) expression primes transendothelial migration of human mesenchymal stem cells. Proc Natl Acad Sci U S A. 2011;108:2258–63. doi: 10.1073/pnas.1018064108. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Schenk S, Mal N, Finan A, Zhang M, Kiedrowski M, Popovic Z, et al. Monocyte chemotactic protein-3 is a myocardial MSC homing factor. Stem Cells. 2007;25:245–51. doi: 10.1634/stemcells.2006-0293. [PubMed] [CrossRef] [Google Scholar]
- Shi M, Li J, Liao L, Chen B, Li B, Chen L, et al. Regulation of CXCR4 expression in human mesenchymal stem cells by cytokine treatment: role in homing efficiency in NOD/SCID mice. Haematologica. 2007;92:897–904. doi: 10.3324/haematol.10669. [PubMed] [CrossRef] [Google Scholar]
- Potapova IA, Brink PR, Cohen IS, Doronin SV. Culturing of human mesenchymal stem cells as three-dimensional aggregates induces functional expression of CXCR4 that regulates adhesion to endothelial cells. J Biol Chem. 2008;283:13100–7. doi: 10.1074/jbc.M800184200. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Caplan A, Correa D. The MSC: an injury drugstore. Cell Stem Cell. 2011;9:11–5. doi: 10.1016/j.stem.2011.06.008. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Stappenbeck TS, Miyoshi H. The role of stromal stem cells in tissue regeneration and wound repair. Science. 2009;324:1666–9. doi: 10.1126/science.1172687. [PubMed] [CrossRef] [Google Scholar]
- Furlani D, Ugurlucan M, Ong L, Bieback K, Pittermann E, Westien I, et al. Is the intravascular administration of mesenchymal stem cells safe? Mesenchymal stem cells and intravital microscopy. Microvasc Res. 2009;77:370–6. doi: 10.1016/j.mvr.2009.02.001. [PubMed] [CrossRef] [Google Scholar]
- Sharma S, Krishan A. Hematopoietic Stem Cells. In: Krishan A, Krishnamurthy H, Totey S, editors. Applications of flow cytometry in stem cell research and tissue regeneration. New Jersey: Wiley-Blackwell; 2010. pp. 103–14. [Google Scholar]
- Radley JM, Ellis S, Palatsides M, Williams B, Bertoncello I. Ultrastructure of primitive hematopoietic stem cells isolated using probes of functional status. Exp Hematol. 1999;27:365–9. doi: 10.1016/S0301-472X(98)00017-4. [PubMed] [CrossRef] [Google Scholar]
- Lee RH, Pulin AA, Seo MJ, Kota DJ, Ylostalo J, Larson BL, et al. Intravenous hMSCs improve myocardial infarction in mice because cells embolized in lung are activated to secrete the anti-inflammatory protein TSG-6. Cell Stem Cell. 2009;5:54–63. doi: 10.1016/j.stem.2009.05.003. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Deak E, Rüster B, Keller L, Eckert K, Fichtner I, Seifried E, et al. Suspension medium influences interaction of mesenchymal stromal cells with endothelium and pulmonary toxicity after transplantation in mice. Cytotherapy. 2010;12:260–4. doi: 10.3109/14653240903401840. [PubMed] [CrossRef] [Google Scholar]
- Fischer UM, Harting MT, Jimenez F, Monzon-Posadas WO, Xue H, Savitz SI, et al. Pulmonary passage is a major obstacle for intravenous stem cell delivery: the pulmonary first-pass effect. Stem Cells Dev. 2009;18(5):683–92. doi: 10.1089/scd.2008.0253. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Kerkelä E, Hakkarainen T, Mäkelä T, Raki M, Kambur O, Kilpinen L, et al. Transient proteolytic modification of mesenchymal stromal cells increases lung clearance rate and targeting to injured tissue. Stem Cells Transl Med. 2013;2(7):510–20. doi: 10.5966/sctm.2012-0187. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Walczak P, Zhang J, Gilad AA, Kedziorek DA, Ruiz-Cabello J, Young RG, et al. Dual-modality monitoring of targeted intraarterial delivery of mesenchymal stem cells after transient ischemia. Stroke. 2008;39:1569–74. doi: 10.1161/STROKEAHA.107.502047. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Mäkelä T, Takalo R, Arvola O, Haapanen H, Yannopoulos F, Blanco R, et al. Safety and biodistribution study of bone marrow-derived mesenchymal stromal cells and mononuclear cells and the impact of the administration route in an intact porcine model. Cytotherapy. 2015;17:392–402. doi: 10.1016/j.jcyt.2014.12.004. [PubMed] [CrossRef] [Google Scholar]
- Toupet K, Maumus M, Luz-Crawford P, Lombardo E, Lopez-Belmonte J, van Lent P, et al. Survival and biodistribution of xenogenic adipose mesenchymal stem cells is not affected by the degree of inflammation in arthritis. PLoS One. 2015;10:e0114962. doi: 10.1371/journal.pone.0114962. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Tögel F, Yang Y, Zhang P, Hu Z, Westenfelder C. Bioluminescence imaging to monitor the in vivo distribution of administered mesenchymal stem cells in acute kidney injury. Am J Physiol Renal Physiol. 2008;295:F315–21. doi: 10.1152/ajprenal.00098.2008. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Morigi M, De Coppi P. Cell therapy for kidney injury: different options and mechanisms—mesenchymal and amniotic fluid stem cells. Nephron Exp Nephrol. 2014;126:59. doi: 10.1159/000360667. [PubMed] [CrossRef] [Google Scholar]
- Toma C, Wagner WR, Bowry S, Schwartz A, Villanueva F. Fate of culture-expanded mesenchymal stem cells in the microvasculature: in vivo observations of cell kinetics. Circ Res. 2009;104(3):398–402. doi: 10.1161/CIRCRESAHA.108.187724. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Cui LL, Kerkelä E, Bakreen A, Nitzsche F, Andrzejewska A, Nowakowski A, et al. The cerebral embolism evoked by intraaterial delivery of allogeneic bone marrow mesenchymal stem cells in rats is related to cell dose and infusion velocity. Stem Cell Res Ther. 2015;6:11. doi: 10.1186/scrt544. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Nemeth K, Leelahavanichkul A, Yuen PS, Mayer B, Parmelee A, Doi K, et al. Bone marrow stromal cells attenuate sepsis via prostaglandin E(2)-dependent reprogramming of host macrophages to increase their interleukin-10 production. Nat Med. 2009;15:42–9. doi: 10.1038/nm.1905. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Chiesa S, Morbelli S, Morando S, Massollo M, Marini C, Bertoni A, et al. Mesenchymal stem cells impair in vivo T-cell priming by dendritic cells. Proc Natl Acad Sci U S A. 2011;108:17384–9. doi: 10.1073/pnas.1103650108. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Akiyama K, Chen C, Wang D, Xu X, Qu C, Yamaza T, et al. Mesenchymal stem cell-induced immunoregulation involves Fas ligand/Fas-mediated T cell apoptosis. Cell Stem Cell. 2012;10(5):544–55. doi: 10.1016/j.stem.2012.03.007. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Choi H, Lee RH, Bazhanov N, Oh JY, Prockop DJ. Anti-inflammatory protein TSG-6 secreted by activated MSCs attenuates zymosan-induced mouse peritonitis by decreasing TLR2/NF-?B signaling in resident macrophages. Blood. 2011;118:330–8. doi: 10.1182/blood-2010-12-327353. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Kim J, Hematti P. Mesenchymal stem celleducated macrophages: a novel type of alternatively activated macrophages. Exp Hematol. 2009;37:1445–53. doi: 10.1016/j.exphem.2009.09.004. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Maggini J, Mirkin G, Bognanni I, Holmberg J, Piazzón IM, Nepomnaschy I, et al. Mouse bone marrow-derived mesenchymal stromal cells turn activated macrophages into a regulatory-like profile. PLoS One. 2010;5(2):e9252. doi: 10.1371/journal.pone.0009252. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Francois M, Romieu-Mourez R, Li M, Galipeau J. Human MSC suppression correlates with cytokine induction of indoleamine 2,3-dioxygenase and bystander M2 macrophage differentiation. Mol Ther. 2011;20:187–95. doi: 10.1038/mt.2011.189. [PubMed] [CrossRef] [Google Scholar]
- Sasaki M, Abe R, Fujita Y, Ando S, Inokuma D, Shimizu H. Mesenchymal stem cells are recruited into wounded skin and contribute to wound repair by transdifferentiation into multiple skin cell type. J Immunol. 2008;180:2581–7. doi: 10.4049/jimmunol.180.4.2581. [PubMed] [CrossRef] [Google Scholar]
- Le Blanc K, Mougiakanos D. Multipotent mesenchymal stromal cells and the innate immune system. Nat Rev Immunol. 2012;12:383–96. doi: 10.1038/nri3209. [PubMed] [CrossRef] [Google Scholar]
- Nauta AJ, Fibbe WE. Immunomodulatory properties of mesenchymal stromal cells. Blood. 2007;110:3499–506. doi: 10.1182/blood-2007-02-069716. [PubMed] [CrossRef] [Google Scholar]
- von Bahr L, Batsis I, Moll G, Hägg M, Szakos A, Sundberg B, et al. Analysis of tissues following mesenchymal stromal cell therapy in humans indicates limited long-term engraftment and no ectopic tissue formation. Stem Cells. 2012;30:1575–8. doi: 10.1002/stem.1118. [PubMed] [CrossRef] [Google Scholar]
- Segers VFM, Van Riet I, Andries LJ, Lemmens K, Demolder MJ, De Becker AJML, et al. Mesenchymal stem cell adhesion to cardiac microvascular endothelium: activators and mechanisms. Am J Physiol Heart Circ Physiol. 2006;290:H1370–7. doi: 10.1152/ajpheart.00523.2005. [PubMed] [CrossRef] [Google Scholar]
- Ip JE, Wu Y, Huang J, Zhang L, Pratt RE, Dzau VJ. Mesenchymal stem cells use integrin beta1 not CXC chemokine receptor 4 for myocardial migration and engraftment. Mol Biol Cell. 2007;18:2873–82. doi: 10.1091/mbc.E07-02-0166. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Wu Y, Zhao RC. The role of chemokines in mesenchymal stem cell homing to myocardium. Stem Cell Rev. 2012;8:243–50. doi: 10.1007/s12015-011-9293-z. [PubMed] [CrossRef] [Google Scholar]
- Zhang M, Mal N, Kiedrowski M, Chacko M, Askari AT, Popovic ZB, et al. SDF-1 expression by mesenchymal stem cells results in trophic support of cardiac myocytes after myocardial infarction. FASEB J. 2007;21:3197–207. doi: 10.1096/fj.06-6558com. [PubMed] [CrossRef] [Google Scholar]
- Belema-Bedada F, Uchida S, Martire A, Kostin S, Braun T. Efficient homing of multipotent adult MSCs depends on FROUNT-mediated clustering of CCR2. Cell Stem Cell. 2008;2:566–75. doi: 10.1016/j.stem.2008.03.003. [PubMed] [CrossRef] [Google Scholar]
- Wang Y, Zhang D, Ashraf M, Zhao T, Huang W, Ashraf A, et al. Combining neuropeptide Y and mesenchymal stem cells reverses remodeling after myocardial infarction. Am J Physiol Heart Circ Physiol. 2010;298:H275–86. doi: 10.1152/ajpheart.00765.2009. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Laurila JP, Laatikainen L, Castellone MD, Trivedi P, Heikkila J, Hinkkanen A, et al. Human embryonic stem cell-derived mesenchymal stromal cell transplantation in a rat hind limb injury model. Cytotherapy. 2009;11:726–37. doi: 10.3109/14653240903067299. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Jasmin, Jelicks LA, Tanowitz HB, Peters VM, Mendez-Otero R, de Carvalho AC C, et al. Molecular imaging, biodistribution and efficacy of mesenchymal bone marrow cell therapy in a mouse model of Chagas disease. Microbes Infect. 2014;16:923–35. doi: 10.1016/j.micinf.2014.08.016. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Reinders ME, de Fijter JW, Roelofs H, Bajema IM, de Vries DK, Schaapherder AF, et al. Autologous bone marrow-derived mesenchymal stromal cells for the treatment of allograft rejection after renal transplantation: results of a phase I study. Stem Cells Transl Med. 2013;2:107–11. doi: 10.5966/sctm.2012-0114. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Tan J, Wu W, Xu X, Liao L, Zheng F, Messinger S, et al. Induction therapy with autologous mesenchymal stem cells in living-related kidney transplants: a randomized controlled trial. JAMA. 2012;307:1169–77. doi: 10.1001/jama.2012.316. [PubMed] [CrossRef] [Google Scholar]
- Humphreys BD, Bonventre JV. Mesenchymal stem cells in acute kidney injury. Annu Rev Med. 2008;59:311–25. doi: 10.1146/annurev.med.59.061506.154239. [PubMed] [CrossRef] [Google Scholar]
- Gooch A, Doty J, Flores J, Swenson L, Toegel FE, Reiss GR, et al. Initial report on a phase I clinical trial: prevention and treatment of post-operative acute kidney injury with allogeneic mesenchymal stem cells in patients who require on-pump cardiac surgery. Cell Ther Transplant. 2008;1:e.000028.01. [Google Scholar]
- Sugimoto H, Mundel TM, Sund M. Bone-marrow-derived stem cells repair basement membrane collagen defects and reverse genetic kidney disease. Proc Natl Acad Sci U S A. 2006;103:7321–6. doi: 10.1073/pnas.0601436103. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Morigi M, Imberti B, Zoja C, Corna D, Tomasoni S, Abbate M, et al. Mesenchymal stem cells are renotropic, helping to repair the kidney and improve function in acute renal failure. J Am Soc Nephrol. 2004;15(7):1794–804. doi: 10.1097/01.ASN.0000128974.07460.34. [PubMed] [CrossRef] [Google Scholar]
- Imberti B, Morigi M, Tomasoni S, Rota C, Corna D, Longaretti L, et al. Insulin-like growth factor-1 sustains stem cell mediated renal repair. J Am Soc Nephrol. 2007;18:2921–8. doi: 10.1681/ASN.2006121318. [PubMed] [CrossRef] [Google Scholar]
- Bruno S, Grange C, Deregibus MC, Calogero RA, Saviozzi S, Collino F, et al. Mesenchymal stem cell-derived microvesicles protect against acute tubular injury. J Am Soc Nephrol. 2009;20:1053–67. doi: 10.1681/ASN.2008070798. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Briquet A, Grégoire C, Comblain F, Servais L, Zeddou M, Lechanteur C, et al. Human bone marrow, umbilical cord or liver mesenchymal stromal cells fail to improve liver function in a model of CCl4-induced liver damage in NOD/SCID/IL-2R?(null) mice. Cytotherapy. 2014;16:1511–8. doi: 10.1016/j.jcyt.2014.07.003. [PubMed] [CrossRef] [Google Scholar]
- Zhang S, Lv C, Yang X, Han Z, Zhang S, Zhang J, et al. Corticosterone mediates the inhibitory effect of restraint stress on the migration of mesenchymal stem cell to carbon tetrachloride-induced fibrotic liver by downregulating CXCR4/7 expression. Stem Cells Dev. 2015;24(5):587–96. doi: 10.1089/scd.2014.0243. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Li Q, Zhou X, Shi Y, Li J, Zheng L, Cui L, et al. In vivo tracking and comparison of the therapeutic effects of MSCs and HSCs for liver injury. PLoS One. 2013;8(4) doi: 10.1371/journal.pone.0062363. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Parekkadan B, Upadhyay R, Dunham J, Iwamoto Y, Mizoguchi E, Mizoguchi A, et al. Bone marrow stromal cell transplants prevent experimental enterocolitis and require host CD11b?+?splenocytes. Gastroenterology. 2011;140:966–75. doi: 10.1053/j.gastro.2010.10.013. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Wu M, Ji S, Xiao S, Kong Z, Fang H, Zhang Y, et al. JAM-A promotes wound healing by enhancing both homing and secretion activities of mesenchymal stem cells. Clin Sci (Lond). 2015. [Epub ahead of print]. [PubMed]
- Kidd S, Spaeth E, Dembinski JL, Dietrich M, Watson K, Klopp A, et al. Direct evidence of mesenchymal stem cell tropism for tumor and wounding microenvironments using in vivo bioluminescent imaging. Stem Cells. 2009;27:2614. doi: 10.1002/stem.187. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Fathke C, Wilson L, Hutter J, Kapoor V, Smith A, Hocking A, et al. Contribution of bone marrow-derived cells to skin: collagen deposition and wound repair. Stem Cells. 2004;22:812. doi: 10.1634/stemcells.22-5-812. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Wu Y, Chen L, Scott PG, Tredget EE. Mesenchymal stem cells enhance wound healing through differentiation and angiogenesis. Stem Cells. 2007;25:2648. doi: 10.1634/stemcells.2007-0226. [PubMed] [CrossRef] [Google Scholar]
- Wu J, Sun Z, Sun H-S, Wu J, Weisel RD, Keating A, et al. Intravenously administered bone marrow cells migrate to damaged brain tissue and improve neural function in ischemic rats. Cell Transplant. 2008;16:993–1005. doi: 10.3727/000000007783472435. [PubMed] [CrossRef] [Google Scholar]
- Yilmaz G, Vital S, Yilmaz CE, Stokes KY, Alexander JS, Granger DN. Selectin-mediated recruitment of bone marrow stromal cells in the postischemic cerebral microvasculature. Stroke. 2011;42:806–11. doi: 10.1161/STROKEAHA.110.597088. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Wei L, Fraser JL, Lu ZY, Hu X, Yu SP. Transplantation of hypoxia preconditioned bone marrow mesenchymal stem cells enhances angiogenesis and neurogenesis after cerebral ischemia in rats. Neurobiol Dis. 2012;46:635–45. doi: 10.1016/j.nbd.2012.03.002. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Constantin G, Marconi S, Rossi B, Angiari S, Calderan L, Anghileri E, et al. Adipose-derived mesenchymal stem cells ameliorate chronic experimental autoimmune encephalomyelitis. Stem Cells. 2009;27:2624–35. doi: 10.1002/stem.194. [PubMed] [CrossRef] [Google Scholar]
- Simmons PJ, Przepiorka D, Thomas ED, Torok-Storb B. Host origin of marrow stromal cells following allogeneic bone marrow transplantation. Nature. 1987;328:429–32. doi: 10.1038/328429a0. [PubMed] [CrossRef] [Google Scholar]
- Cilloni D, Carlo-Stella C, Falzetti F, Sammarelli G, Regazzi E, Colla S, et al. Limited engraftment capacity of bone marrow-derived mesenchymal cells following T-cell-depleted hematopoietic stem cell transplantation. Blood. 2000;96:3637–43. [PubMed] [Google Scholar]
- Rieger K, Marinets O, Fietz T, Körper S, Sommer D, Mücke C, et al. Mesenchymal stem cells remain of host origin even a long time after allogeneic peripheral blood stem cell or bone marrow transplantation. Exp Hematol. 2005;33:605–11. doi: 10.1016/j.exphem.2005.02.004. [PubMed] [CrossRef] [Google Scholar]
- Rombouts WJ, Ploemacher RE. Primary murine MSC show highly efficient homing to the bone marrow but lose homing ability following culture. Leukemia. 2003;17:160–70. doi: 10.1038/sj.leu.2402763. [PubMed] [CrossRef] [Google Scholar]
- Follenzi A, Raut S, Merlin S, Sarkar R, Gupta S. Role of bone marrow transplantation for correcting hemophilia A in mice. Blood. 2012;119:5532–42. doi: 10.1182/blood-2011-07-367680. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Otsuru S, Gordon PL, Shimono K, Jethva R, Marino R, Phillips CL, et al. Transplanted bone marrow mononuclear cells and MSCs impart clinical benefit to children with osteogenesis imperfecta through different mechanisms. Blood. 2012;120:1933–41. doi: 10.1182/blood-2011-12-400085. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Sémont A, Mouiseddine M, François A, Demarquay C, Mathieu N, Chapel A, et al. Mesenchymal stem cells improve small intestinal integrity through regulation of endogenous epithelial cell homeostasis. Cell Death Differ. 2010;17:952–61. doi: 10.1038/cdd.2009.187. [PubMed] [CrossRef] [Google Scholar]
- Fakhrejahani E, Toi M. Tumor angiogenesis: pericytes and maturation are not to be ignored. J Oncol. 2012;2012:261750. doi: 10.1155/2012/261750. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- D’souza N, Burns JS, Grisendi G, Candini O, Veronesi E, Piccinno S, et al. MSC and tumors: homing, differentiation, and secretion influence therapeutic potential. Adv Biochem Eng Biotechnol. 2013;130:209–66. [PubMed] [Google Scholar]
- Beckermann BM, Kallifatidis G, Groth A, Frommhold D, Apel A, Mattern J, et al. VEGF expression by mesenchymal stem cells contributes to angiogenesis in pancreatic carcinoma. Br J Cancer. 2008;99:622–31. doi: 10.1038/sj.bjc.6604508. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Alieva M, Bagó JR, Aguilar E, Soler-Botija C, Vila OF, Molet J, et al. Glioblastoma therapy with cytotoxic mesenchymal stromal cells optimized by bioluminescence imaging of tumor and therapeutic cell response. PLoS One. 2012;7:e35148. doi: 10.1371/journal.pone.0035148. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Knoop K, Schwenk N, Schmohl K, Müller A, Zach C, Cyran C, et al. Mesenchymal stem cell (MSC)-mediated, tumor stroma-targeted radioiodine therapy of metastatic colon cancer using the sodium iodide symporter as theranostic gene. J Nucl Med. 2015;pii:jnumed.114.146662. [PubMed]
- Xu S, Menu E, De Becker A, Van Camp B, Vanderkerken K, Van Riet I. Bone marrow-derived mesenchymal stromal cells are attracted by multiple myeloma cell-produced chemokine CCL25 and favor myeloma cell growth in vitro and in vivo. Stem Cells. 2012;30:266–79. doi: 10.1002/stem.787. [PubMed] [CrossRef] [Google Scholar]
- Duan X, Guan H, Cao Y, Kleinerman ES. Murine bone marrow-derived mesenchymal stem cells as vehicles for interleukin-12 gene delivery into Ewing sarcoma tumors. Cancer. 2009;115:13–22. doi: 10.1002/cncr.24013. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Kidd S, Spaeth E, Watson K, Burks J, Lu H, Klopp A, et al. Origins of the tumor microenvironment: quantitative assessment of adipose-derived and bone marrow-derived stroma. PLoS One. 2012;7:e30563. doi: 10.1371/journal.pone.0030563. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Grisendi G, Bussolari R, Veronesi E, Piccinno S, Burns JS, De Santis G, et al. Understanding tumor-stroma interplays for targeted therapies by armed mesenchymal stromal progenitors: the Mesenkillers. Am J Cancer Res. 2011;1:787–805. [PMC free article] [PubMed] [Google Scholar]
- Suzuki K, Sun R, Origuchi M, Kanehira M, Takahata T, Itoh J, et al. Mesenchymal stromal cells promote tumor growth through the enhancement of neovascularization. Mol Med. 2011;17:579–87. doi: 10.2119/molmed.2010.00157. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Yu B, Zhang X, Li X. Exosomes derived from mesenchymal stem cells. Int J Mol Sci. 2014;15:4142–57. doi: 10.3390/ijms15034142. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Akyurekli C, Le Y, Richardson RB, Fergusson D, Tay J, Allan DS. A systematic review of preclinical studies on the therapeutic potential of mesenchymal stromal cell-derived microvesicles. Stem Cell Rev. 2015;11:150–60. doi: 10.1007/s12015-014-9545-9. [PubMed] [CrossRef] [Google Scholar]
- Zhang Y, Chopp M, Meng Y, Katakowski M, Xin H, Mahmood A, et al. Effect of exosomes derived from multipluripotent mesenchymal stromal cells on functional recovery and neurovascular plasticity in rats after traumatic brain injury. J Neurosurg. 2015;16:1–12. [PMC free article] [PubMed] [Google Scholar]
- Xin H, Li Y, Liu Z, Wang X, Shang X, Cui Y, et al. MiR-133b promotes neural plasticity and functional recovery after treatment of stroke with multipotent mesenchymal stromal cells in rats via transfer of exosome-enriched extracellular particles. Stem Cells. 2013;31:2737–46. doi: 10.1002/stem.1409. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Tomasoni S, Longaretti L, Rota C, Morigi M, Conti S, Gotti E, et al. Transfer of growth factor receptor mRNA via exosomes unravels the regenerative effect of mesenchymal stem cells. Stem Cells Dev. 2013;22:772–80. doi: 10.1089/scd.2012.0266. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Kordelas L, Rebmann V, Ludwig AK, Radtke S, Ruesing J, Doeppner TR, et al. MSC-derived exosomes: a novel tool to treat therapy-refractory graft-versus-host disease. Leukemia. 2014;28:970–3. [PubMed] [Google Scholar]
- Spaggiari GM, Capobianco A, Becchetti S, Mingari MC, Moretta L. Mesenchymal stem cell-natural killer cell interactions: evidence that activated NK cells are capable of killing MSCs, whereas MSCs can inhibit IL-2-induced NK-cell proliferation. Blood. 2006;107:1484–90. doi: 10.1182/blood-2005-07-2775. [PubMed] [CrossRef] [Google Scholar]
- Giuliani M, Bennaceur-Griscelli A, Nanbakhsk A, Oudrhiri N, Chouaib S, Azzarone B, et al. TLR ligands stimulation protects MSC from NK killing. Stem Cells. 2014;32:290–300. doi: 10.1002/stem.1563. [PubMed] [CrossRef] [Google Scholar]
- Reinders ME, Hoogduijn MJ. NK cells and MSCs: possible implications for MSC therapy in renal transplantation. J Stem Cell Res Ther. 2014;4:10000166. doi: 10.4172/2157-7633.1000166. [PMC free article] [PubMed] [CrossRef] [Google Scholar]
- Moll G, Rasmusson-Duprez I, Von Bahr L, Conolly-Andersen AM, Elgue G, Funke L, et al. Are therapeutic human mesenchymal stromal cells compatible with human blood. Stem Cells. 2012;30:1565–74. doi: 10.1002/stem.1111. [PubMed] [CrossRef] [Google Scholar]
Articles from Stem Cell Research & Therapy are provided here courtesy of BioMed Central
- Published in Corporate News / Blog
The FDA Vs Stem Cell Treatments In The US
The FDA Vs Stem Cell Treatments In The US
Stem cell therapy has become a popular type of treatment for a number of conditions, including to assist in the regeneration of injured tissue or diseases cells in the human body. This therapy is classified as regenerative medicine. Several advancements have already been made in this medical field, and many had experienced positive results when they underwent stem cell therapy to assist in the treatment of degenerative conditions.
While stem cell therapy has been proven to provide an effective protocol in the recovery of stroke, neurological issues, and even to assist in repairing the damage that was dealt by a heart attack, recent reports have caused many to view these treatments in a negative perspective. This is due to the warnings that were recently issued by the FDA. Even though some have experienced side-effects due to the use of stem cell therapy, it is important to note that there are facilities within the US that can provide professional services that help to minimize the risk of these adverse events.
The FDA On Stem Cell Therapy In The United States
The Food and Drug Administration of the United States is involved in the analyzing and approval of medications and medical procedures that may be offered to patients in the country. To date, the FDA has shown some interest in stem cell therapy, providing approval for several clinical trials that looked at how effective this treatment is in providing a regenerating effect on the impact that certain conditions has in the human body. Only one particular treatment that involves stem cell therapy has been officially approved by the Food and Drug Administration, however.
Within the last few months, we have seen several concerning publications made by the FDA regarding stem cell therapies, reducing the hope that some patients might have gained.
On the 25th of June, 2019, the FDA published a statement that described a permanent injunction that will occur with the many stem cell clinics in the country. The FDA has stated that the action taken from their side is to assist in providing a layer of protection for patients, due to the risks involved with undergoing treatments that utilize products that have not gone through any type of approval process.
The specific company that was targeted in this statement and by the actions that were taken by the FDA is known as US Stem Cell Clinic LLC. The court ruled in favor of the FDA and US government earlier in June, finding the defendants, being US Stem Cell Clinic LLC, guilty of the claims that were made against them.
An earlier publication also confirmed that the Federal court has decided to rule the misbranding of the stem cell products released by the US Stem Cell Clinics company as a violation of laws that have been implemented to protect the people of the country.
News publications have also announced that the regulations and guidelines that have now been set out by the FDA might cause a significant decline in the availability of stem cell treatments to patients in the United States. The companies that have been targeted through these particular actions that the FDA has taken are primarily those that have been marketing products that have not gone through approval phases – and these products have been found to put the patient’s health at risk.
The Role Of Stem Cell Therapy In Disease
While some companies have been found guilty of using unapproved and even misleading products on patients, promoted as stem cell therapy, it is important to consider the reality of the situation as well. Stem cell therapy often referred to simply as Cell therapy, has been proven as a successful regime in the treatment of several conditions. Many of the conditions that have shown improvement with the use of this therapy were previously considered difficult, or sometimes even impossible, to effectively treat.
By 2012, the Worldwide Network for Blood and Marrow Transplantation, also known as the WBMT, announced that a total of one million stem cell therapy procedures had been done throughout the world. This was a significant milestone, and these procedures have helped to save a countless number of lives. Just in this report, the WBMT refers back to a case of Marta, a girl from Madrid, who had received stem cell therapy at a young age. This treatment was provided to Marta after she was diagnosed with Leukemia. In 2002, Marta was able to overcome Leukemia, thanks to the cell therapy that was provided to her.
While lymphoma and leukemia are the conditions where patients most frequently seek a clinic that can perform stem cell therapy on them, there are many other conditions that are treated with this procedure today. The perfection of the therapy has led to a treatment that can assist in reducing the effects of over 70 diseases that may otherwise have a significant impact on the human body.
GSCG Expands To Cancun, Meets The Demands Of US Patients
The Global Stem Cells Group had recently announced the opening of a new office that they have decided to establish in Cancun, Mexico. The decision was driven by the increasing demand for cell therapy procedures by patients in the United States and by the fact that there is currently a lack of facilities that are able to provide these individuals with the professional and quality services needed. Doctors specializing in regenerative medicine are also having a hard time fulfilling the needs of patients who wish to consider cell therapy as a potential treatment option.
This is not the only local office that GSCG now operates in Cancun, as the company now has two facilities within this particular location. These offices include a stem cells laboratory, as well as a general medical facility.
Over the years, GSCG has become an established name in the stem cell field, providing thousands of doctors the opportunity to offer cell therapy as a treatment option for patients with degenerative diseases. The use of this therapy may assist in the regeneration of damaged and diseased tissues in the patient’s body.
Even though a previous office had established a presence for GSCG in Cancun, it should be noted that the company announced that the opening of the new office means they have now officially created a permanent presence for the brand in the area.
The GSCG’s new facility in Cancun is equipped with the latest advancements that have been made in the field and provide quality services that ensure patients have a trusted medical facility where their patients can undergo cell therapy as part of a regenerative medicine treatment plan.
At the moment, doctors who are in the field of regenerative medicine within the United States can turn to this facility to offer their patients an additional treatment option, apart from the standard pharmaceutical protocols that have been established. The stem cell laboratory can assist in the analyzing of patient data and also in the process of finding a matching donor for the person who is in need of stem cell treatment.
GSCG’s facility is able to assist with the culturing of expanded autonomous cells, as well as other types of cells that may be utilized in the process of stem cell therapy. A detailed approach is taken, considering the age of the donor, as this would also provide an overview of the autonomous stem cell age. The patient being treated is also taken into close consideration, as there are different methods of introducing these stem cells to the body for enhanced efficacy.
A more aggressive method is often needed in those patients with conditions such as autism, Alzheimer’s disease, and kidney-related conditions. Other conditions can also be treated, such as pulmonary diseases, Parkinson’s disease. In cases of a stroke, the vascular arteries, along with the carotid artery, is targeted with the therapy for a more effective approach to assisting with the regeneration of damaged structures in the patient’s brain. This ultimately leads to a significant improvement in the delivery of stem cells to the patient’s body, which could enhance the overall efficacy expressed by the treatment.
When these procedures are provided to the patient through the GSCG, an appropriate ICU unit is made available to ensure the patient is provided with adequate care during the operation and recovery of the procedure.
The utilization of recent technological advancements at laboratories facilitated by GSCG can also assist in providing a more economical approach to cell therapy. The utilization of allogenic stem cell procedures can be addressed using techniques that will essentially offer a reduction in the costs involved.
Planar technologies may be utilized when the demand is less than one billion cells per annum, which can significantly reduce the costs of the process and, in turn, make these treatments available to patients at a lower price.
The utilization of autologous stem cell transplantation also offers an opportunity for patients to undergo stem cell therapy when a sibling does not provide an ideal match. Cells can be cultivated from the bone marrow and treated with specific drugs in order to yield targeted results. In the case of malignant cells in the body, bone marrow can be treated with monoclonal antibodies or cytotoxic drugs to target such cells when transplanted back into the patient.
Appropriate storage solutions are also taken into consideration to assist with the cultivation process, and to reduce the risk of enzymatic treatment leads to the differentiation of cells into other types. The culture condition becomes crucial for the expansion of stem cells. A PA6 conditioned medium, along with an enclosure of semi-permeable hollow fiber membranes, is one example of how the laboratory can keep certain stem cells from differentiating, while also assuring successful cultivation and expansion.
The GSCG’s Role In The New FDA Warnings
The fact that the FDA has announced the warnings against stem cell therapy products and had even taken action against a relatively popular company in the United States have now caused a limitation in access to these treatments. With a seizure of the services provided by companies that have been affected by the FDA’s actions and the knowledge of the misleading marketing information that was used in the campaigns initiated by these companies, patients might not be sure where to turn to right now.
The same problem is faced by many doctors in the field of regenerative treatments and medicine. Doctors in these fields need to ensure a facility they utilize to allow patients to undergo a treatment like a cell therapy can offer a safe procedure.
This is where the GSCG comes into the bigger picture. With the establishment of a permanent presence in Mexico, doctors are now given access to a facility that has an established history for providing quality stem cell therapy services to patients with qualifying diseases. The company is not only trusted but have been performing stem cell therapy procedures on patients that had led to successful results.
These treatments may help to assist in repairing neurological problems caused by disorders like Parkinson’s disease, stroke, or an injury to the spinal cord. They may also offer new hope to people with diabetes, heart disease, and even those who had previously suffered damage to their cardiovascular system due to a heart attack.
Conclusion
Even though recent publications from the FDA has caused concerns to be raised regarding stem cell therapy, many patients are still interested in undergoing these treatment procedures. A significant number of studies have provided evidence on the efficacy of treating tissue damage caused by various conditions through stem cell therapy. Patients interested in undergoing stem cell treatment are now advised to turn their interest to Stem Cells Centers in Cancun and related regions, where highly specialized and experienced doctors are able to provide a professional service.
- Published in Corporate News / Blog
![Secondary-Diabetes[1]](https://stemcellcenter.net/wp-content/uploads/2020/02/Secondary-Diabetes1-1-460x260_c.png)





![article1-1[1]](https://stemcellcenter.net/wp-content/uploads/2019/11/article1-11-460x260_c.jpg)
![article2-1[1]](https://stemcellcenter.net/wp-content/uploads/2019/11/article2-11-460x260_c.jpg)
