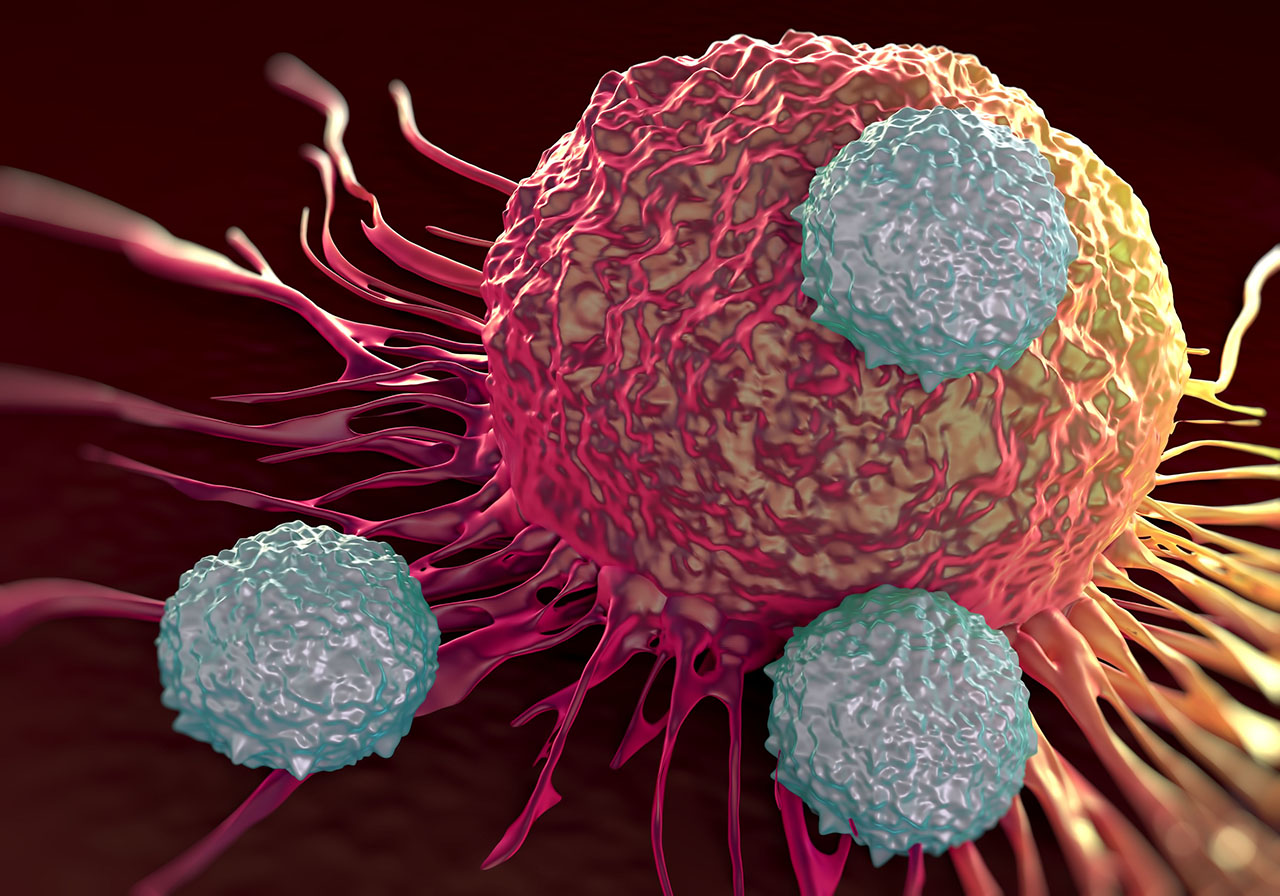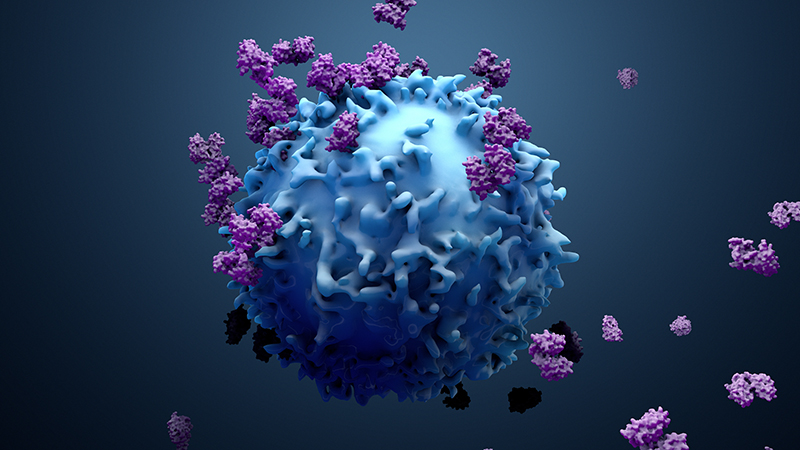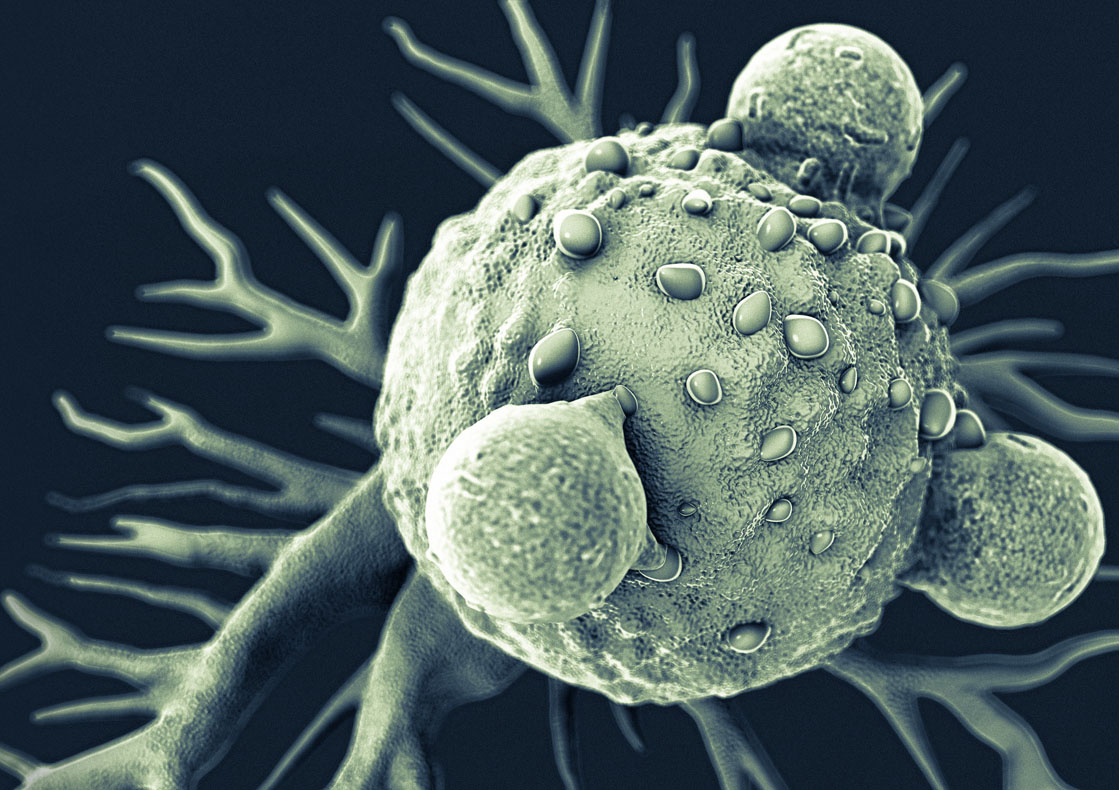
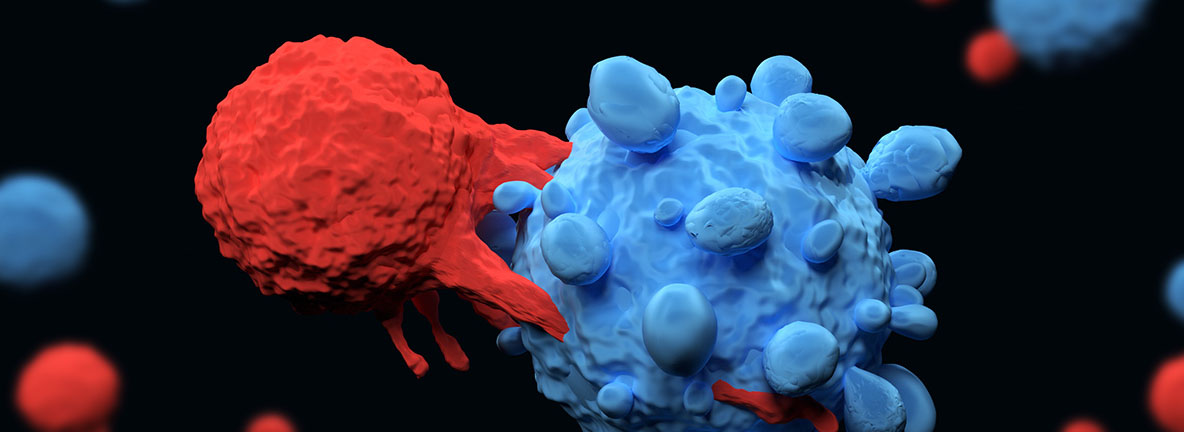
Immunotherapy CAR T stands for Chimeric Antigen Receptor T-cell therapy, which is a form of immunotherapy.
With CAR T therapy, your immune system’s T cells are modified to recognize and destroy cancer cells.
CAR T therapy is an option if your primary and secondary treatments failed to eliminate your cancer, or if your cancer has returned after these treatments.
Before receiving CAR T therapy, you will undergo low-dose chemotherapy in order to prepare for treatment. CAR T may also be an option for patients not eligible to receive a stem cell transplant.
CAR T cells are the equivalent of “giving patients a “living drug” As its name implies, the backbone of CAR T-cell therapy is T cells, which are often called the workhorses of the immune system because of their critical role in orchestrating the immune response and killing cells infected by pathogens.
The therapy requires drawing blood from patients and separating out the T cells. Next, using a disarmed virus, the T cells are genetically engineered to produce receptors on their surface called chimeric antigen receptors, or CARs.
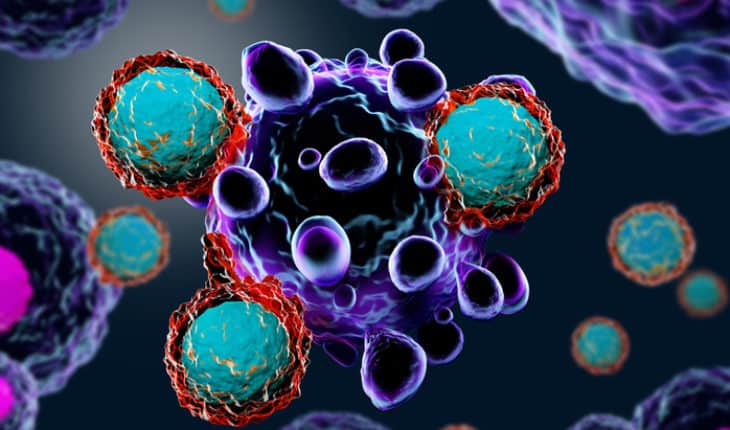
These receptors are synthetic molecules, they don’t exist naturally. These special receptors allow the T cells to recognize and attach to a specific protein, or antigen, on tumor cells. The CAR T cell therapies furthest along in development target an antigen found on B cells called CD19.
Once the collected T cells have been engineered to express the antigen-specific CAR, they are “expanded” in the laboratory into the hundreds of millions.
The final step is the infusion of the CAR T cells into the patient (which is preceded by a “lymphodepleting” chemotherapy regimen.
If all goes as planned, the engineered cells further multiply in the patient’s body and, with guidance from their engineered receptor, recognize and kill cancer cells that harbor the antigen on their surfaces.